-
question_answer1)
Which of the following pairs give positive Tollen's test?
A)
Glucose, sucrose done
clear
B)
Glucose, fructose done
clear
C)
Hexanal, acetophenone done
clear
D)
Fructose, sucrose done
clear
View Solution play_arrow
-
question_answer2)
The correct statement about the following disaccharide is
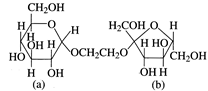
A)
Ring (1) is pyranose with \[\alpha \]-glycosidic link done
clear
B)
Ring (1) is furanose with \[\alpha \]-glycosidic link done
clear
C)
Ring (2) is furanose with \[\alpha \]-glycosidic link done
clear
D)
Ring (2) is pyranose with \[\beta \]-glycosidic link done
clear
View Solution play_arrow
-
question_answer3)
The number of disulphide linkages present in insulin is
A)
4 done
clear
B)
3 done
clear
C)
2 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer4)
Which one of the following is not correct?
A)
d(-) Fructose exists in furanose structure done
clear
B)
d(+) Glucose exists in pyranose structure done
clear
C)
In sucrose the two monosaccharides are held together by peptide linkage done
clear
D)
Maltose is a reducing sugar done
clear
View Solution play_arrow
-
question_answer5)
| Which of the statements about "Denaturation" given below are correct? |
| (1) Denaturation of proteins causes loss of secondary and tertiary structure of the proteins. |
| (2) Denaturation leads to the conversion of double strand of DNA into single strand. |
| (3) Denaturation affects primary structure which gets distorted. |
A)
(2) and (3) done
clear
B)
(1) and (3) done
clear
C)
(1) and (2) done
clear
D)
(1), (2) and (3) done
clear
View Solution play_arrow
-
question_answer6)
Which of the following is not a fat soluble vitamin?
A)
Vitamin B complex done
clear
B)
Vitamin D done
clear
C)
Vitamin E done
clear
D)
Vitamin A done
clear
View Solution play_arrow
-
question_answer7)
How many pairs in the gene are needed to code for the enzyme, lysozyme, containing 129 amino acids found in egg white?
A)
\[3\text{ }\times \text{ }129\] base pairs done
clear
B)
\[\left( 3\times 129 \right)+\left( 3\times 3 \right)=396\]base pairs done
clear
C)
\[\left( 3\times 129 \right)+\left( 3\times 2 \right)=393\]base pairs done
clear
D)
4\[\times \]129 base pairs done
clear
View Solution play_arrow
-
question_answer8)
| Consider the following statements about haemoglobin. |
| (1) It has four separate peptide subunits: Two identical \[\alpha \]-chains and two identical \[\beta \]-chains each bonded to haeme. |
| (2) In CO poisoning, since much of the haemoglobin is ied up with CO, \[{{O}_{2}}\] transport to the tissues is inhibited. |
| (3) Normal haemoglobin found in red blood cells has a glutamic acid residue. The correct statements are |
A)
I and II done
clear
B)
II and III done
clear
C)
I and III done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer9)
Which of the following statements is incorrect?
A)
Vitamins are included in diet because they are not synthesized in the human body done
clear
B)
A person with diabetes mellitus suffers from hypoglycemia done
clear
C)
Most vitamins function as coenzymes done
clear
D)
Hypoglycemia can affect the brain due to low blood sugar level done
clear
View Solution play_arrow
-
question_answer10)
X controls the process of burning of fats, proteins and carbohydrates and liberate energy in the body X is
A)
thyroxine done
clear
B)
adrenaline done
clear
C)
cortisone done
clear
D)
insulin done
clear
View Solution play_arrow
-
question_answer11)
Peptides are formed by the joining of amino acids through amide linkage. Which of the following statement is not true in this respect?
A)
Amide groups do not contribute in the hydrogen bonding interactions done
clear
B)
\[p\pi \] resonance stabilizes the amide linkages done
clear
C)
Amide groups are more resistant to hydrolysis than the similar ester groups done
clear
D)
Stable conformations of peptides are restricted to those having planar amide groups done
clear
View Solution play_arrow
-
question_answer12)
Identity X, Y and Z from the information given below: X = A chiral amino acid which loses its chirality on treatment with lithium aluminum hydride. Y= the decarboxylation product of a-amino (secondary) acid, \[{{C}_{5}}{{H}_{9}}{{O}_{2}}N\]. Z = It is formed when A (\[{{C}_{3}}{{H}_{4}}{{O}_{3}}\]) is treated with (i) \[N{{H}_{3}}\](ii) \[NaB{{H}_{4}}\] [A forms oxime, gives iodoform test and liberate \[C{{O}_{2}}\] with \[NaHC{{O}_{3}}\]].
A)
B)
C)
D)
View Solution play_arrow
-
question_answer13)
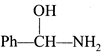
\[Ph-CH=0\xrightarrow{HCN+KOH}X\xrightarrow{PC{{l}_{5}}}Y\xrightarrow[(ii){{H}_{2}}{{O}^{\oplus }}]{(i)N{{H}_{3}}}Z\]will be
A)
B)
C)
D)
View Solution play_arrow
-
question_answer14)
Select the correct statement about the following disaccharide?
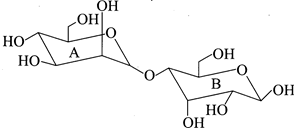
A)
Ring A is an acetal in the\[\beta \]-configuration; ring B is a hemiacetal in the \[\alpha \]-configuration done
clear
B)
Ring A is an acetal in the \[\alpha \]-configuration; ring B is a hemiacetal in the \[\beta \]-configuration done
clear
C)
Ring A is a hemiacetal in the \[\alpha \]-configuration; ring B is an acetal in the \[\beta \]-configuration done
clear
D)
Ring A is a hemiacetal in the \[\beta \]-configuration; ring B is an acetal in the \[\alpha \]-configuration. done
clear
View Solution play_arrow
-
question_answer15)
The correct statement about the following disaccharide is
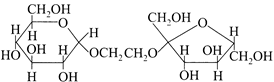
A)
Ring (I) is pyranose with \[\alpha \]-glycosidic link done
clear
B)
Ring (I) is furanose with \[\alpha \]-glycosidic link done
clear
C)
Ring (II) is furanose with \[\alpha \]-glycosidic link done
clear
D)
Ring (II) is pyranose with \[\beta \]-glycosidic link done
clear
View Solution play_arrow
-
question_answer16)
Which two of the following compounds are reduced to the same chiral alditol by sodium borohydride?
A)
II and III done
clear
B)
I and II done
clear
C)
III and IV done
clear
D)
II and IV done
clear
View Solution play_arrow
-
question_answer17)
Identify the correct set of stereochemical relationships amongst the following monosaccharides I-IV
A)
I and II are anomers; III and IV are epimers done
clear
B)
I and II are epimers; II and IV are anomers done
clear
C)
I and III are anomers; I and II are epimers done
clear
D)
I and II are epimers; III and IV are anomers done
clear
View Solution play_arrow
-
question_answer18)
Which of the following indicates the presence of 5 -OH groups in glucose?
A)
Penta-acetyl derivative of glucose done
clear
B)
Cyanohydrin formation of glucose done
clear
C)
Reaction with Fehling's solution done
clear
D)
Reaction with Tollens reagent done
clear
View Solution play_arrow
-
question_answer19)
Acetone may be obtained from starch by the action of
A)
acid done
clear
B)
bacteria done
clear
C)
oxidizing agent done
clear
D)
none of these done
clear
View Solution play_arrow
-
question_answer20)
Two hexoses form the same osazone, find the correct statement about these hexoses.
A)
Both of them must be aldoses done
clear
B)
They are epimers at C-3 done
clear
C)
The carbon atoms 1 and 2 in both have the same configuration done
clear
D)
The carbon atoms 3, 4 and 5 in both have the same configuration done
clear
View Solution play_arrow
-
question_answer21)
What is the number of carbon at6ms in the ketone used to prepare \[\varepsilon \]-aminovaleric acid, a monomer for nylon category?
View Solution play_arrow
-
question_answer22)
Consider the given structure,
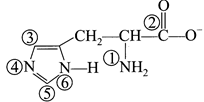
When this molecule is kept in acidic medium, x and y positions gets protonated readily. What is the sum of x and Y?
View Solution play_arrow
-
question_answer23)
Consider an amylase chain of 4000 glucose units. How many cleavage are required to lower the average length to 400 units?
View Solution play_arrow
-
question_answer24)
Some of the pair of monosaccharides are given below:
| (I) Allose, atrose |
(II) Glucose, fructose |
| (III) Glucose, mannose |
(IV) Mannose, fructose |
| (V) Glucose, indose |
(VI) Galactose, talose |
| (VII) Galactose, glucose |
(VIII) Indose, glucose |
How many of the above pairs are C-2 epimers?
View Solution play_arrow
-
question_answer25)
A decapeptide (Mol. Wt. 796) on complete hydrolysis gives glycine (Mol. Wt. 75), alanine and phenylalanine. Glycine contributes 47.0% to the total weight of the hydrolysed products. The number of glycine units present in the decapeptide is
View Solution play_arrow

 done
clear
done
clear


