-
question_answer1)
The following reaction is known as \[{{C}_{2}}{{H}_{5}}OH+SOC{{l}_{2}}\xrightarrow{\text{Pyridine}}{{C}_{2}}{{H}_{5}}Cl+S{{O}_{2}}+HCl\] [AIIMS 2002]
A)
Kharasch effect done
clear
B)
Darzen's procedure done
clear
C)
Williamson's synthesis done
clear
D)
Hunsdiecker synthesis reaction done
clear
View Solution play_arrow
-
question_answer2)
What is the main product of the reaction between 2-methyl propene with HBr [RPMT 2002]
A)
1-bromo butane done
clear
B)
1-bromo-2 methyl propane done
clear
C)
2-bromo butane done
clear
D)
2-bromo-2 methyl propane done
clear
View Solution play_arrow
-
question_answer3)
Halogenation of alkanes is [KCET 2002]
A)
A reductive process done
clear
B)
An oxidative process done
clear
C)
An isothermal process done
clear
D)
An indothermal process done
clear
View Solution play_arrow
-
question_answer4)
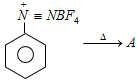
In the above process product A is [Kerala (Engg.) 2002]
A)
Fluorobenzene done
clear
B)
Benzene done
clear
C)
1, 4-difluorobenzene done
clear
D)
1, 3-difluorobenzene done
clear
View Solution play_arrow
-
question_answer5)
Silver acetate\[+\ B{{r}_{2}}\xrightarrow{\text{C}{{\text{S}}_{\text{2}}}}\]. The main product of this reaction is [Kurukshetra CET 2002]
A)
\[C{{H}_{3}}-Br\] done
clear
B)
\[C{{H}_{3}}COI\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer6)
Diazonium salts
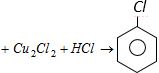
the reaction is known as [Kerala (Med.) 2002]
A)
Chlorination done
clear
B)
Sandmeyer's reaction done
clear
C)
Perkin reaction done
clear
D)
Substitution reaction done
clear
View Solution play_arrow
-
question_answer7)
When ethyl alcohol\[({{C}_{2}}{{H}_{5}}OH)\]reacts with thionyl chloride, in the presence of pyridine, the product obtained is [AIIMS; CBSE PMT 2001]
A)
\[C{{H}_{3}}C{{H}_{2}}Cl+HCl\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}Cl+HCl+S{{O}_{2}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}Cl+{{H}_{2}}O+S{{O}_{2}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}Cl+HCl+S{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer8)
Preparation of alkyl halides in laboratory is least preferred by [DPMT 2000]
A)
Treatment of alcohols done
clear
B)
Addition of hydrogen halides to alkenes done
clear
C)
Halide exchange done
clear
D)
Direct halogenation of alkanes done
clear
View Solution play_arrow
-
question_answer9)
Which of the following organic compounds will give a mixture of 1-chlorobutane and 2-chlorobutane on chlorination [CPMT 2001]
A)
\[C{{H}_{3}}-\underset{C{{H}_{3}}\,\,\,\,\,\,\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{CH-CH}}\,}}\,=C{{H}_{2}}\] done
clear
B)
\[HC\equiv C-\overset{H\,\,}{\mathop{\overset{|\,\,\,\,\,\,}{\mathop{C=}}\,}}\,C{{H}_{2}}\] done
clear
C)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\] done
clear
D)
\[C{{H}_{2}}=CH-C{{H}_{2}}-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer10)
The chlorobenzene is generally obtained from a corresponding diazonium salt by reacting it with [MP PMT 2000]
A)
\[C{{u}_{2}}C{{l}_{2}}\] done
clear
B)
\[CuS{{O}_{4}}\] done
clear
C)
\[Cu\] done
clear
D)
\[Cu(N{{H}_{3}})_{4}^{2+}\] done
clear
View Solution play_arrow
-
question_answer11)
Decreasing order of reactivity of\[HX\]in the reaction \[ROH+HX\to RX+{{H}_{2}}O\] [RPET 2000; AIIMS 1983; MP PET 1996]
A)
\[HI>HBr>HCl>HF\] done
clear
B)
\[HBr>HCl>HI>HF\] done
clear
C)
\[HCl>HBr>HI>HF\] done
clear
D)
\[HF>HBr>HCl>HI\] done
clear
View Solution play_arrow
-
question_answer12)
The product of the following reaction : \[C{{H}_{2}}=CH-CC{{l}_{3}}+HBr\] [RPET 2000]
A)
\[C{{H}_{3}}-CH(Br)-CC{{l}_{3}}\] done
clear
B)
\[C{{H}_{2}}(Br)-C{{H}_{2}}-CC{{l}_{3}}\] done
clear
C)
\[BrC{{H}_{2}}-CHCl-CHC{{l}_{2}}\] done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-CC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer13)
Chlorobenzene is prepared commercially by [JIPMER 2000; CPMT 1976; Pb. CET 2002]
A)
Raschig process done
clear
B)
Wurtz Fitting reaction done
clear
C)
Friedel-Craft?s reaction done
clear
D)
Grignard reaction done
clear
View Solution play_arrow
-
question_answer14)
In methyl alcohol solution, bromine reacts with ethylene to yield \[BrC{{H}_{2}}C{{H}_{2}}OC{{H}_{3}}\] in addition to 1, 2-dibromoethane because [Pb. PMT 1998]
A)
The ion formed initially may react with \[B{{r}^{-}}\] or \[C{{H}_{3}}OH\] done
clear
B)
The methyl alcohol solvates the bromine done
clear
C)
The reaction follows Markownikoff's rule done
clear
D)
This is a free-radical mechanism done
clear
View Solution play_arrow
-
question_answer15)
\[{{C}_{3}}{{H}_{8}}+C{{l}_{2}}\xrightarrow{\text{Light}}{{C}_{3}}{{H}_{7}}Cl+HCl\] is an example of which of the following types of reactions [AFMC 1997; CPMT 1999]
A)
Substitution done
clear
B)
Elimination done
clear
C)
Addition done
clear
D)
Rearrangement done
clear
View Solution play_arrow
-
question_answer16)
Which of the following would be produced when acetylene reacts with HCl [MH CET 1999]
A)
\[C{{H}_{3}}C{{H}_{2}}Cl\] done
clear
B)
\[C{{H}_{3}}CHC{{l}_{2}}\] done
clear
C)
\[CHCl=CHCl\] done
clear
D)
\[C{{H}_{2}}=CHCl\] done
clear
View Solution play_arrow
-
question_answer17)
\[R-OH+HX\to R-X+{{H}_{2}}O\] In the above reaction, the reactivity of different alcohols is [CPMT 1997]
A)
Tertiary > Secondary > Primary done
clear
B)
Tertiary < Secondary < Primary done
clear
C)
Tertiary < Secondary > Primary done
clear
D)
Secondary < Primary < Tertiary done
clear
View Solution play_arrow
-
question_answer18)
\[{{C}_{6}}{{H}_{6}}+C{{l}_{2}}\xrightarrow{UV\text{ Light}}\] Product. In above reaction product is [CPMT 1997]
A)
\[CC{{l}_{3}}CHO\] done
clear
B)
\[{{C}_{6}}{{H}_{6}}C{{l}_{6}}\] done
clear
C)
\[{{C}_{6}}{{H}_{12}}C{{l}_{6}}\] done
clear
D)
\[{{C}_{6}}{{H}_{9}}C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer19)
Benzene reacts with chlorine to form benzene hexachloride in presence of [MP PET 1999]
A)
Nickel done
clear
B)
\[AlC{{l}_{3}}\] done
clear
C)
Bright sunlight done
clear
D)
Zinc done
clear
View Solution play_arrow
-
question_answer20)
The final product obtained by distilling ethyl alcohol with the excess of chlorine and \[Ca{{(OH)}_{2}}\] is [MP PET 1996]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[CC{{l}_{3}}CHO\] done
clear
C)
\[CHC{{l}_{3}}\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer21)
When ethyl alcohol and KI reacted in presence of \[N{{a}_{2}}C{{O}_{3}},\] yellow crystals of...... are formed [AFMC 1989]
A)
\[CH{{I}_{3}}\] done
clear
B)
\[C{{H}_{3}}I\] done
clear
C)
\[C{{H}_{2}}{{I}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}I\] done
clear
View Solution play_arrow
-
question_answer22)
In preparation of \[CHC{{l}_{3}}\] from ethanol and bleaching powder, the latter provides [BHU 1986]
A)
\[Ca{{(OH)}_{2}}\] done
clear
B)
\[C{{l}_{2}}\] done
clear
C)
Both A and B done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer23)
Which one of the following processes does not occur during formation of \[CHC{{l}_{3}}\] from \[{{C}_{2}}{{H}_{5}}OH\] and bleaching powder [DPMT 1984]
A)
Hydrolysis done
clear
B)
Oxidation done
clear
C)
Reduction done
clear
D)
Chlorination done
clear
View Solution play_arrow
-
question_answer24)
Which of the following is obtained when chloral is boiled with NaOH [CBSE PMT 1991; RPMT 1999]
A)
\[C{{H}_{3}}Cl\] done
clear
B)
\[CHC{{l}_{3}}\] done
clear
C)
\[CC{{l}_{4}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer25)
Chloroform can be obtained from [MNR 1986]
A)
Methanol done
clear
B)
Methanal done
clear
C)
Propanol-1 done
clear
D)
Propanol-2 done
clear
View Solution play_arrow
-
question_answer26)
Chlorine reacts with ethanol to give [MP PMT 1989; CPMT 1997; KCET 1998; JIPMER 1999]
A)
Ethyl chloride done
clear
B)
Chloroform done
clear
C)
Acetaldehyde done
clear
D)
Chloral done
clear
View Solution play_arrow
-
question_answer27)
On heating diethyl ether with conc. HI, 2 moles of which of the following is formed [IIT-JEE 1983; MP PET 1990; EAMCET 1990; AFMC 1993; JIPMER 2001]
A)
Ethanol done
clear
B)
Iodoform done
clear
C)
Ethyl iodide done
clear
D)
Methyl iodide done
clear
View Solution play_arrow
-
question_answer28)
Lucas reagent is [MP PMT 1996; MP PET 1992, 95; CPMT 1986, 89; AIIMS 1980; Kurukshetra CEE 2002]
A)
Concentrated \[HCl+\text{anhydrous }ZnC{{l}_{2}}\] done
clear
B)
Dilute \[HCl+\text{ hydrated }ZnC{{l}_{2}}\] done
clear
C)
Concentrated \[HN{{O}_{3}}+\text{ anhydrous }ZnC{{l}_{2}}\] done
clear
D)
Concentrated \[HCl+\text{ anhydrous }MgC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer29)
Which compound does not form iodoform with alkali and iodine [IIT-JEE 1985]
A)
Acetone done
clear
B)
Ethanol done
clear
C)
Diethyl ketone done
clear
D)
Isopropyl alcohol done
clear
View Solution play_arrow
-
question_answer30)
Which compound gives yellow ppt. with iodine and alkali [IIT-JEE 1984]
A)
2-hydroxy propane done
clear
B)
Acetophenone done
clear
C)
Methyl acetone done
clear
D)
Acetamide done
clear
View Solution play_arrow
-
question_answer31)
Acetone reacts with \[{{I}_{2}}\] in presence of \[NaOH\] to form [MP PMT 1992]
A)
\[{{C}_{2}}{{H}_{5}}I\] done
clear
B)
\[{{C}_{2}}{{H}_{4}}{{I}_{2}}\] done
clear
C)
\[CH{{I}_{3}}\] done
clear
D)
\[C{{H}_{3}}I\] done
clear
View Solution play_arrow
-
question_answer32)
Ethanol is converted into ethyl chloride by reacting with [MP PET 1991; MP PMT 1990; BHU 1997]
A)
\[C{{l}_{2}}\] done
clear
B)
\[SOC{{l}_{2}}\] done
clear
C)
HCl done
clear
D)
NaCl done
clear
View Solution play_arrow
-
question_answer33)
\[{{C}_{6}}{{H}_{5}}Cl\] prepared by aniline with [IIT-JEE 1984]
A)
HCl done
clear
B)
\[C{{u}_{2}}C{{l}_{2}}\] done
clear
C)
\[C{{l}_{2}}\] in presence of anhydrous \[AlC{{l}_{3}}\] done
clear
D)
\[HN{{O}_{2}}\] and then heated with \[C{{u}_{2}}C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer34)
The starting substance for the preparation of \[C{{H}_{3}}I\] is [CPMT 1975]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[C{{H}_{3}}CHO\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}CO\] done
clear
View Solution play_arrow
-
question_answer35)
A Grignard's reagent may be made by reacting magnesium with [CPMT 1973, 83, 84]
A)
Methyl amine done
clear
B)
Diethyl ether done
clear
C)
Ethyl iodide done
clear
D)
Ethyl alcohol done
clear
View Solution play_arrow
-
question_answer36)
Which of the following is responsible for iodoform reaction [CPMT 1980; RPMT 1997]
A)
Formalin done
clear
B)
Methanol done
clear
C)
Acetic acid done
clear
D)
Ethanol done
clear
View Solution play_arrow
-
question_answer37)
When a solution of sodium chloride containing ethyl alcohol is electrolysed, it forms
A)
Ethyl alcohol done
clear
B)
Chloral done
clear
C)
Chloroform done
clear
D)
Acetaldehyde done
clear
View Solution play_arrow
-
question_answer38)
Which reagent cannot be used to prepare an alkyl halide from an alcohol [CPMT 1989, 94]
A)
\[HCl+ZnC{{l}_{2}}\] done
clear
B)
NaCl done
clear
C)
\[PC{{l}_{5}}\] done
clear
D)
\[SOC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer39)
Ethyl benzoate reacts with \[PC{{l}_{5}}\]to give [KCET 2003]
A)
\[{{C}_{2}}{{H}_{5}}Cl+{{C}_{6}}{{H}_{5}}COCl+POC{{l}_{3}}+HCl\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}Cl+{{C}_{6}}{{H}_{5}}COCl+POC{{l}_{3}}\] done
clear
C)
\[C{{H}_{3}}COCl+{{C}_{6}}{{H}_{5}}COCl+POC{{l}_{3}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}Cl+{{C}_{6}}{{H}_{5}}COOH+POC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer40)
On treatment with chlorine in presence of sunlight, toluene giv.es the product [Orissa JEE 2003; MH CET 1999, 2002]
A)
o-chloro toluene done
clear
B)
2, 5-dichloro toluene done
clear
C)
p-chloro toluene done
clear
D)
Benzyl chloride done
clear
View Solution play_arrow
-
question_answer41)
When chlorine is passed through warm benzene in presence of the sunlight, the product obtained is [KCET 2003]
A)
Benzotrichloride done
clear
B)
Chlorobenzene done
clear
C)
Gammexane done
clear
D)
DDT done
clear
View Solution play_arrow
-
question_answer42)
Which of the following acids adds to propene in the presence of peroxide to give anti-Markownikoff?s product [MP PET 2003]
A)
\[HF\] done
clear
B)
\[HCl\] done
clear
C)
\[HBr\] done
clear
D)
\[HI\] done
clear
View Solution play_arrow
-
question_answer43)
Propene on treatment with HBr gives [CPMT 1986]
A)
Isopropyl bromide done
clear
B)
Propyl bromide done
clear
C)
1, 2-dibromoethane done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer44)
The catalyst used in Raschig's process is
A)
\[LiAl{{H}_{4}}\] done
clear
B)
Copper chloride done
clear
C)
Sunlight done
clear
D)
\[{\text{Ethanol}}/{Na}\;\] done
clear
View Solution play_arrow
-
question_answer45)
The compound formed on heating chlorobenzene with chloral in the presence of concentrated sulphuric acid, is [AIEEE 2004]
A)
Freon done
clear
B)
DDT done
clear
C)
Gammexene done
clear
D)
Hexachloroethane done
clear
View Solution play_arrow
-
question_answer46)
Acetone is mixed with bleaching powder to give [AFMC 2004]
A)
Chloroform done
clear
B)
Acetaldehyde done
clear
C)
Ethanol done
clear
D)
Phosgene done
clear
View Solution play_arrow
-
question_answer47)
Which of the following compounds gives trichloromethane on distilling with bleaching powder [KCET 2004; EAMCET 1986]
A)
Methanal done
clear
B)
Phenol done
clear
C)
Ethanol done
clear
D)
Methanol done
clear
View Solution play_arrow
-
question_answer48)
The product formed on reaction of ethyl alcohol with bleaching powder is [Orissa JEE 2004; DPMT 1978; AIIMS 1991]
A)
\[CHC{{l}_{3}}\] done
clear
B)
\[CC{{l}_{3}}CHO\] done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}CHO\] done
clear
View Solution play_arrow
-
question_answer49)
Ethylene reacts with bromine to form [Pb. CET 2000]
A)
Chloroethane done
clear
B)
Ethylene dibromide done
clear
C)
Cyclohexane done
clear
D)
1-bromo propane done
clear
View Solution play_arrow
-
question_answer50)
Best method of preparing alkyl chloride is [MH CET 2004]
A)
\[ROH+SOC{{l}_{2}}\xrightarrow{\ \ \ \ \ \ \ \ \ }\] done
clear
B)
\[ROH+PC{{l}_{5}}\xrightarrow{\ \ \ \ \ \ \ \ \ \ \ \ }\] done
clear
C)
\[ROH+PC{{l}_{3}}\xrightarrow{\ \ \ \ \ \ \ \ \ \ \ \ }\] done
clear
D)
\[ROH+HCl\xrightarrow{\text{anhy}\text{. }ZnC{{l}_{2}}}\] done
clear
View Solution play_arrow
-
question_answer51)
DDT is prepared by reacting chlorobenzene with [BHU 1998, 2005]
A)
\[CC{{l}_{4}}\] done
clear
B)
\[CC{{l}_{3}}-CHO\] done
clear
C)
\[CHC{{l}_{3}}\] done
clear
D)
Ethane done
clear
View Solution play_arrow
-
question_answer52)
Which compound needs chloral in its synthesis [Pb. PET 2003]
A)
D. D. T. done
clear
B)
Gammexane done
clear
C)
Chloroform done
clear
D)
Michler?s Ketone done
clear
View Solution play_arrow
-
question_answer53)
To get DDT, chlorobenzene has to react with which of the following compounds in the presence of concentrated sulphuric acid [KCET (Engg/Med.) 2001]
A)
Trichloroethane done
clear
B)
Dichloroacetone done
clear
C)
Dichloroacetaldehyde done
clear
D)
Trichloroacetaldehyde done
clear
View Solution play_arrow
-
question_answer54)
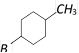
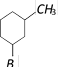
What will be the product in the following reaction
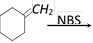
[BHU 2005]
A)
B)
C)
D)
View Solution play_arrow
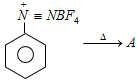 In the above process product A is [Kerala (Engg.) 2002]
In the above process product A is [Kerala (Engg.) 2002] 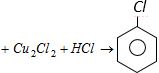 the reaction is known as [Kerala (Med.) 2002]
the reaction is known as [Kerala (Med.) 2002] ![]() [BHU 2005]
[BHU 2005] 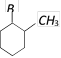 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
