A) iso-propyl
B) Triphenylmethyl cation
C) Ethyl cation
D) \[\pi \]-propyl cation
Correct Answer: B
Solution :
Stability of carbonium ions. tertiary alkyl > secondary alkyl > primary alkyl > methyl. More the number of alkyl groups, the greater the dispersal of positive charge and therefore, more the stability of carbonium ion is observed.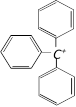 triphenylmethyl cation Dispersal of positive charge increases with the increase in the number of benzene ring.
triphenylmethyl cation Dispersal of positive charge increases with the increase in the number of benzene ring.
You need to login to perform this action.
You will be redirected in
3 sec
