Answer:
The following features observed in photoelectric effect support the photon picture of e. m. radiation: (i) The maximum kinetic energy of emitted electrons is independent of the intensity of incident radiation. (ii) For each photo emitter, there exists a threshold frequency (of incident light) below which no emission takes place. (iii) Photoelectric emission is an instantaneous process. According to Einstein's photoelectric equation \[{{K}_{\max }}=hv-{{W}_{0}}\] 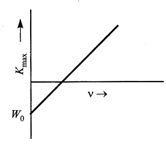 So the graph between \[{{K}_{\max }}\]and \[\text{v}\] is a straight line as shown in Fig. (i) Slope of \[{{K}_{\max }}-v\] graph \[=\frac{\Delta {{K}_{\max }}}{\Delta v}=h\] \[\therefore \] Slope of \[{{K}_{\max }}-v\] graph gives the value of Planck's constant. (ii) Intercept on the negative \[{{K}_{\max }}\]axis \[={{W}_{0}}\] \[\therefore \]Intercept on the negative \[{{K}_{\max }}\] axis gives the value of work function. Energy of incident photon \[=\]Maximum K.E of Photoelectron \[+\]Work function of metal or \[hv=\frac{1}{2}m\upsilon _{\max }^{2}+{{W}_{0}}\] At threshold frequency \[{{v}_{0}}\], no K.E. is given to the photoelectron, so \[h{{v}_{0}}={{W}_{0}}\] \[[{{K}_{\max }}=\frac{1}{2}m\upsilon _{\max }^{2}=0]\] or \[{{v}_{0}}=\frac{{{W}_{0}}}{h}\].
So the graph between \[{{K}_{\max }}\]and \[\text{v}\] is a straight line as shown in Fig. (i) Slope of \[{{K}_{\max }}-v\] graph \[=\frac{\Delta {{K}_{\max }}}{\Delta v}=h\] \[\therefore \] Slope of \[{{K}_{\max }}-v\] graph gives the value of Planck's constant. (ii) Intercept on the negative \[{{K}_{\max }}\]axis \[={{W}_{0}}\] \[\therefore \]Intercept on the negative \[{{K}_{\max }}\] axis gives the value of work function. Energy of incident photon \[=\]Maximum K.E of Photoelectron \[+\]Work function of metal or \[hv=\frac{1}{2}m\upsilon _{\max }^{2}+{{W}_{0}}\] At threshold frequency \[{{v}_{0}}\], no K.E. is given to the photoelectron, so \[h{{v}_{0}}={{W}_{0}}\] \[[{{K}_{\max }}=\frac{1}{2}m\upsilon _{\max }^{2}=0]\] or \[{{v}_{0}}=\frac{{{W}_{0}}}{h}\].
You need to login to perform this action.
You will be redirected in
3 sec
