A) \[-N=N-\]
B) \[-NO\]
C) \[-N{{O}_{2}}\]
D) \[-N{{H}_{2}}\]
Correct Answer: D
Solution :
A dye molecule is made up of two different part (i) Chromophore (ii) Auxochrome. The groups which produce colour in a compound i.e., chromophores are usual unsaturated groups like nitro, nitroso, azo, azoxy, carbonyl and olefinic bonds Auxochrome are usually acidic / basic functional group like \[-OH,\] \[-COOH,\] \[-S{{O}_{3}}H,\] \[-N{{H}_{2}},\] \[-NHR,\] \[-N{{R}_{2}}\]. A compound containing only a chromophore may be coloured material but not a dye. For example azo benzene is red coloured but not a dye. Where as para amino azobenzene (aniline yellow) is a dye.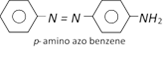
You need to login to perform this action.
You will be redirected in
3 sec
