Answer:
Case - 1 Water Case - 2 Un known liquid \[{{h}_{1}}=13.6\text{ }cm\] \[{{h}_{2}}=1\text{ }cm\] \[{{P}_{1}}=P\] \[{{P}_{2}}=P\] \[{{d}_{1}}=1g\text{ }c{{m}^{3}}\] \[{{d}_{2}}=?\] \[{{g}_{1}}=g\] \[{{g}_{2}}=g\] 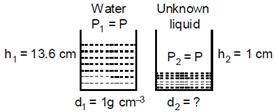 We know, \[P=hdg\] Both the liquids are exerting same pressures. So, \[{{P}_{1}}={{P}_{2}}\] \[{{h}_{1}}{{d}_{1}}{{g}_{1}}={{h}_{2}}{{d}_{2}}{{g}_{2}}\] \[\Rightarrow \] \[{{h}_{1}}{{d}_{1}}={{h}_{2}}{{d}_{2}}\] (At a given place\[{{g}_{1}}={{g}_{2}}\]) \[\Rightarrow \]\[{{d}_{2}}=\frac{{{h}_{1}}{{d}_{1}}}{{{h}_{2}}}=\frac{13.6\times 1}{1}=13.6\,g\,c{{m}^{-3}}\] \[13.6\text{ }g\text{ }c{{m}^{3}}\]is the density of mercury, So, the unknown liquid is mercury.
We know, \[P=hdg\] Both the liquids are exerting same pressures. So, \[{{P}_{1}}={{P}_{2}}\] \[{{h}_{1}}{{d}_{1}}{{g}_{1}}={{h}_{2}}{{d}_{2}}{{g}_{2}}\] \[\Rightarrow \] \[{{h}_{1}}{{d}_{1}}={{h}_{2}}{{d}_{2}}\] (At a given place\[{{g}_{1}}={{g}_{2}}\]) \[\Rightarrow \]\[{{d}_{2}}=\frac{{{h}_{1}}{{d}_{1}}}{{{h}_{2}}}=\frac{13.6\times 1}{1}=13.6\,g\,c{{m}^{-3}}\] \[13.6\text{ }g\text{ }c{{m}^{3}}\]is the density of mercury, So, the unknown liquid is mercury.
You need to login to perform this action.
You will be redirected in
3 sec
