A) \[s{{p}^{3}}{{d}^{3}}\]
B) \[s{{p}^{2}}d\]
C) \[{{d}^{2}}s{{p}^{3}}\]
D) \[s{{p}^{3}}\]
Correct Answer: A
Solution :
The electronic configuration of \[I=[Xe]\ 5{{s}^{2}},5{{p}^{5}}\] hence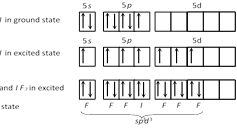 IF7 shows \[s{{p}^{3}}{{d}^{2}}\]hybridization. So, its structure is pentagonal bipyramidal.
IF7 shows \[s{{p}^{3}}{{d}^{2}}\]hybridization. So, its structure is pentagonal bipyramidal.
You need to login to perform this action.
You will be redirected in
3 sec
