A) \[s{{p}^{3}}\]-hybridization and tetrahedral geometry
B) \[s{{p}^{3}}\]-hybridization and distorted tetrahedral geometry
C) \[s{{p}^{2}}\]-hybridization and triangular geometry
D) None of these
Correct Answer: B
Solution :
In the ammonia molecule N atom is \[s{{p}^{3}}-\]hybridized but due to the presence of one lone pair of \[{{e}^{-}}\] (i.e. due to greater \[{{L}_{p}}-{{b}_{p}}\]repulsion) it has distorted tetrahedral (or pyramidal) geometry.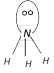
You need to login to perform this action.
You will be redirected in
3 sec
