-
question_answer1)
In the following which bond will be responsible for maximun value of hydrogen bond
A)
\[O-H\] done
clear
B)
\[N-H\] done
clear
C)
\[S-H\] done
clear
D)
\[F-H\] done
clear
View Solution play_arrow
-
question_answer2)
In which of the following hydrogen bond is present
A)
\[{{H}_{2}}\] done
clear
B)
Ice done
clear
C)
Sulphur done
clear
D)
Hydrocarbon done
clear
View Solution play_arrow
-
question_answer3)
In the following which has highest boiling point [MP PMT 1989; RPMT 1997]
A)
\[HI\] done
clear
B)
\[HF\] done
clear
C)
\[HBr\] done
clear
D)
\[HCl\] done
clear
View Solution play_arrow
-
question_answer4)
Which contains hydrogen bond [MP PMT 1989]
A)
\[HF\] done
clear
B)
\[HCl\] done
clear
C)
\[HBr\] done
clear
D)
\[HI\] done
clear
View Solution play_arrow
-
question_answer5)
Contrary to other hydrogen halides, hydrogen fluoride is a liquid because [MP PMT 1990; AMU 1983; EAMCET 1980]
A)
Size of \[F\]atom is small done
clear
B)
\[HF\]is a weak acid done
clear
C)
\[HF\]molecule are hydrogen bonded done
clear
D)
Fluorine is highly reactive done
clear
View Solution play_arrow
-
question_answer6)
In the following which species does not contain \[s{{p}^{3}}\] hybridization [DPMT 1985]
A)
\[SO_{4}^{2-}\] done
clear
B)
\[C{{H}_{4}}\] done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer7)
As a result of \[sp\] hybridization, we get [IIT 1984]
A)
Two mutual perpendicular orbitals done
clear
B)
Two orbitals at \[{{180}^{o}}\] done
clear
C)
Four orbitals in tetrahedral directions done
clear
D)
Three orbitals in the same plane done
clear
View Solution play_arrow
-
question_answer8)
The reason for exceptionally high boiling point of water is [DPMT 1986; NCERT 1976; AMU 1984; EAMCET 1979; MP PMT 1993; AIIMS 1996; KCET 2001; CPMT 2003]
A)
Its high specific heat done
clear
B)
Its high dielectric constant done
clear
C)
Low ionization of water molecules done
clear
D)
Hydrogen bonding in the molecules of water done
clear
View Solution play_arrow
-
question_answer9)
Which concept best explains that o-nitrophenol is more volatile than p-nitrophenol [AIIMS 1980, 82; Kurukshetra CEE 1998; MP PET 2002]
A)
Resonance done
clear
B)
Hyperconjugation done
clear
C)
Hydrogen bonding done
clear
D)
Steric hindrence done
clear
View Solution play_arrow
-
question_answer10)
Which contains strongest \[H-\]bond [IIT 1986; MP PET 1997, 2003; UPSEAT 2001, 03]
A)
\[O-H.....S\] done
clear
B)
\[S-H.....O\] done
clear
C)
\[F-H.....F\] done
clear
D)
\[S-H.....O\] done
clear
View Solution play_arrow
-
question_answer11)
Which of the following compound can form hydrogen bonds [NCERT 1978; MP PMT 1997]
A)
\[C{{H}_{4}}\] done
clear
B)
\[NaCl\] done
clear
C)
\[CHC{{l}_{3}}\] done
clear
D)
\[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer12)
Of the following hydrides which has the lowest boiling point [CBSE PMT 1987]
A)
\[N{{H}_{3}}\] done
clear
B)
\[P{{H}_{3}}\] done
clear
C)
\[Sb{{H}_{3}}\] done
clear
D)
\[As{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer13)
The pairs of bases in \[DNA\] are held together by [NCERT 1978; DPMT 1985; CBSE PMT 1992]
A)
Hydrogen bonds done
clear
B)
Ionic bonds done
clear
C)
Phosphate groups done
clear
D)
Deoxyribose groups done
clear
View Solution play_arrow
-
question_answer14)
Water has high heat of vaporisation due to [AFMC 1982]
A)
Covalent bonding done
clear
B)
\[H-\]bonding done
clear
C)
Ionic bonding done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer15)
In which of the following compounds does hydrogen bonding occur [CBSE PMT 1989]
A)
\[Si{{H}_{4}}\] done
clear
B)
\[LiH\] done
clear
C)
\[HI\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer16)
Which among the following compounds does not show hydrogen bonding [MP PMT 1989]
A)
Chloroform done
clear
B)
Ethyl alcohol done
clear
C)
Acetic acid done
clear
D)
Ethyl ether done
clear
View Solution play_arrow
-
question_answer17)
Acetic acid exists as dimer in benzene due to [CPMT 1982]
A)
Condensation reaction done
clear
B)
Hydrogen bonding done
clear
C)
Presence of carboxyl group done
clear
D)
Presence of hydrogen atom at \[\alpha -\]carbon done
clear
View Solution play_arrow
-
question_answer18)
Which one among the following does not have the hydrogen bond [IIT 1983; MP PMT 1994; UPSEAT 2001]
A)
Phenol done
clear
B)
Liquid \[N{{H}_{3}}\] done
clear
C)
Water done
clear
D)
Liquid \[HCl\] done
clear
View Solution play_arrow
-
question_answer19)
The bond that determines the secondary structure of a protein is [NCERT 1984; MP PET 1996]
A)
Coordinate bond done
clear
B)
Covalent bond done
clear
C)
Hydrogen bond done
clear
D)
Ionic bond done
clear
View Solution play_arrow
-
question_answer20)
\[HCl\] is a gas but \[HF\]is a low boiling liquid. This is because [NCERT 1984; MP PMT 2001]
A)
\[H-F\]bond is strong done
clear
B)
\[H-F\]bond is weak done
clear
C)
Molecules aggregate because of hydrogen bonding done
clear
D)
\[HF\]is a weak acid done
clear
View Solution play_arrow
-
question_answer21)
The relatively high boiling point of \[HF\]is due to [NCERT 1984]
A)
Hydrogen bonding done
clear
B)
Covalent bonding done
clear
C)
Unshared electron pair on \[F\] done
clear
D)
Being a halogen acid done
clear
View Solution play_arrow
-
question_answer22)
Water is liquid due to [MADT Bihar 1983]
A)
Hydrogen bonding done
clear
B)
Covalent bond done
clear
C)
Ionic bond done
clear
D)
Vander Waals forces done
clear
View Solution play_arrow
-
question_answer23)
The maximum possible number of hydrogen bonds in which an \[{{H}_{2}}O\]molecule can participate is [MP PMT 1986; MNR 1991; IIT 1992;MP PET 1999]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer24)
Hydrogen bonding is maximum in [IIT 1987; MP PMT 1991; MP PET 1993, 2001; MNR 1995; CPMT 1999; KCET (Med.) 2002]
A)
Ethanol done
clear
B)
Diethyl ether done
clear
C)
Ethyl chloride done
clear
D)
Triethyl amine done
clear
View Solution play_arrow
-
question_answer25)
The hydrogen bond is strongest in [BHU 1987; CBSE PMT 1990, 92]
A)
Water done
clear
B)
Ammonia done
clear
C)
Hydrogen fluoride done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer26)
The high boiling point of ethanol \[({{78.2}^{o}}C)\] compared to dimethyl ether\[(-{{23.6}^{o}}C)\], though both having the same molecular formulae \[{{C}_{6}}{{H}_{6}}O\], is due to [MP PMT 1993]
A)
Hydrogen bonding done
clear
B)
Ionic bonding done
clear
C)
Coordinate covalent bonding done
clear
D)
Resonance done
clear
View Solution play_arrow
-
question_answer27)
Methanol and ethanol are miscible in water due to [CPMT 1989]
A)
Covalent character done
clear
B)
Hydrogen bonding character done
clear
C)
Oxygen bonding character done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer28)
B.P. of \[{{H}_{2}}O\,({{100}^{o}}C)\] and \[{{H}_{2}}S\,(-{{42}^{o}}C)\] explained by
A)
Vander Waal's forces done
clear
B)
Covalent bond done
clear
C)
Hydrogen bond done
clear
D)
Ionic bond done
clear
View Solution play_arrow
-
question_answer29)
Strength of hydrogen bond is intermediate between [DPMT 1991]
A)
Vander Waal and covalent done
clear
B)
Ionic and covalent done
clear
C)
Ionic and metallic done
clear
D)
Metallic and covalent done
clear
View Solution play_arrow
-
question_answer30)
In which of the following compounds intramolecular hydrogen bond is present [MP PET 1994]
A)
Ethyl alcohol done
clear
B)
Water done
clear
C)
Salicylaldehyde done
clear
D)
Hydrogen sulphide done
clear
View Solution play_arrow
-
question_answer31)
Hydrogen bonding is formed in compounds containing hydrogen and [MP PET 1995]
A)
Highly electronegative atoms done
clear
B)
Highly electropositive atoms done
clear
C)
Metal atoms with d-orbitals occupied done
clear
D)
Metalloids done
clear
View Solution play_arrow
-
question_answer32)
Which of the following compounds in liquid state does not have hydrogen bonding [MP PMT 1996]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer33)
Compounds showing hydrogen bonding among
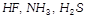
and
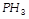
are
A)
B)
C)
D)
All the four done
clear
View Solution play_arrow
-
question_answer34)
The high density of water compared to ice is due to [CBSE PMT 1997; BHU 1999; AFMC 2001]
A)
Hydrogen bonding interactions done
clear
B)
Dipole-dipole interactions done
clear
C)
Dipole-induced dipole interactions done
clear
D)
Induced dipole-induced dipole interactions done
clear
View Solution play_arrow
-
question_answer35)
Ethanol and dimethyl ether form a pair of functional isomers. The boiling point of ethanol is higher than that of dimethyl ether due to the presence of [AIIMS 1998]
A)
Hydrogen bonding in ethanol done
clear
B)
Hydrogen bonding in dimethyl ether done
clear
C)
D)
View Solution play_arrow
-
question_answer36)
Which of the following hydrogen bonds are strongest in vapour phase [AMU 1999]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer37)
Which of the following shows hydrogen bonding [CPMT 2000]
A)
B)
P done
clear
C)
As done
clear
D)
Sb done
clear
View Solution play_arrow
-
question_answer38)
The boiling point of a compound is raised by [DPMT 2001]
A)
Intramolecular hydrogen bonding done
clear
B)
Intermolecular hydrogen bonding done
clear
C)
Covalent bonding done
clear
D)
Ionic covalent done
clear
View Solution play_arrow
-
question_answer39)
The boiling point of water is exceptionally high because [KCET 2001]
A)
Water molecule is linear done
clear
B)
Water molecule is not linear done
clear
C)
There is covalent bond between H and O done
clear
D)
Water molecules associate due to hydrogen bonding done
clear
View Solution play_arrow
-
question_answer40)
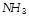
has a much higher boiling point than
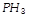
because [UPSEAT 2002; MNR 1994]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer41)
Which one has the highest boiling point [MP PET 2002]
A)
Acetone done
clear
B)
Ethyl alcohol done
clear
C)
Diethyl ether done
clear
D)
Chloroform done
clear
View Solution play_arrow
-
question_answer42)
Which of the following compounds has the highest boiling point [JIPMER 2002]
A)
HCl done
clear
B)
HBr done
clear
C)
D)
View Solution play_arrow
-
question_answer43)
Which of the following has minimum melting point [UPSEAT 2003]
A)
CsF done
clear
B)
HCl done
clear
C)
HF done
clear
D)
LiF done
clear
View Solution play_arrow
-
question_answer44)
Hydrogen bond energy is equal to [UPSEAT 2003]
A)
3 ? 7 kcals done
clear
B)
30 ? 70 kcals done
clear
C)
3 ? 10 kcals done
clear
D)
30 ? 70 kcals done
clear
View Solution play_arrow
-
question_answer45)
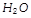
is a liquid while
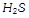
is gas due to [BHU 2003]
A)
Covalent bonding done
clear
B)
Molecular attraction done
clear
C)
H ? bonding done
clear
D)
H ? bonding and molecular attraction done
clear
View Solution play_arrow
-
question_answer46)
H ? bonding is maximum in [BHU 2003]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer47)
Select the compound from the following which dissolves in water [IIT 1980]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer48)
When two ice cubes are pressed over each other, they unit to form one cube. Which of the following force is responsible for holding them together [NCERT 1978]
A)
Vander Waal's forces done
clear
B)
Hydrogen bond formation done
clear
C)
Covalent attraction done
clear
D)
Dipole?dipole attraction done
clear
View Solution play_arrow
-
question_answer49)
Which is the weakest among the following types of bond [NCERT 1979; MADT Bihar 1984]
A)
Ionic bond done
clear
B)
Metallic bond done
clear
C)
Covalent bond done
clear
D)
Hydrogen bond done
clear
View Solution play_arrow
-
question_answer50)
H-bond is not present in [BCECE 2005]
A)
Water done
clear
B)
Glycerol done
clear
C)
Hydrogen fluoride done
clear
D)
Hydrogen Sulphide done
clear
View Solution play_arrow
![]() and
and ![]() are
are ![]() has a much higher boiling point than
has a much higher boiling point than ![]() because [UPSEAT 2002; MNR 1994]
because [UPSEAT 2002; MNR 1994] ![]() is a liquid while
is a liquid while ![]() is gas due to [BHU 2003]
is gas due to [BHU 2003] 