question_answer 1)
Which of the following contains both covalent and ionic bond?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 2)
Which of the following is not a correct statement?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 3)
Main axis of a diatomic molecule is Z. \[A{O}'s\] \[{{p}_{x}}\]and \[{{p}_{y}}\] overlap to form which of the following orbitals?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 4)
The group having triangular planar structures is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
In piperidine
the hybrid state assumed by N is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
The compound\[M{{X}_{4}}\]is tetrahedral. The number of\[\angle XMX\] formed in the compound are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 7)
The correct order of hybridization of the central atom in the following species \[N{{H}_{3}},{{[PtC{{l}_{4}}]}^{2+}},\text{ }PC{{l}_{5}}\] and \[BC{{l}_{3}}\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
The bond dissociation energy of \[B-F\] in\[~B{{F}_{3}}\] is \[646\text{ }kJ\text{ }mo{{l}^{-1}}\] whereas that of \[C-F\] in \[C{{F}_{4}}\] is\[515\text{ }kJ\text{ }mo{{l}^{-1}}\]. The correct reason for higher \[B-F\] bond dissociation energy as compared to that of \[C-F\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
Amongst \[LiCl\],\[RbCl\],\[BeC{{l}_{2}}\], and \[Mg{{C}_{2}}\], the compounds with the greatest and the least ionic character, respectively are:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
Which of these statements is not true?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
Which of the following does not apply to metallic bond?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
In\[PO{{_{4}^{3}}^{-}}\], the formal charge on each oxygen atom and the \[P-O\]bond order respectively are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
Which one of the following formulae does not correctly represent the bonding capacities of the two atoms involved?
A)
B)
C)
D)
View Solution play_arrow
question_answer 14)
The dipole moments of diatomic molecules AB and CD are 10.41D and 10.27 D, respectively while their bond distances are 2.82 and \[2.67\overset{\text{o}}{\mathop{\text{A}}}\,\], respectively. This indicates that
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
Among the following, the species having the smallest bond is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
Which compound exhibits maximum dipole moment among the following?
A)
B)
C)
D)
View Solution play_arrow
question_answer 17)
Molecular AB has a bond length of \[1.61\overset{\text{o}}{\mathop{\text{A}}}\,\]and a dipole moment of 0.38 D. The fractional charge on each atom (absolute magnitude) is: \[(e=4.802\times {{10}^{-10}}esu)\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
In a regular octahedral molecule,\[M{{X}_{6}}\] the number of \[X-M-X\] bonds at \[180{}^\circ \] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 19)
Arrange the following ions in the order of decreasing\[~X-O\] bond length, where X is the central atom
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 20)
Which of the following statements is correct in the context of the allene molecule, \[{{C}_{3}}{{H}_{4}}\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 21)
Which of the following pairs have identical bond order?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 22)
Which of the following molecular orbitals has two nodal planes?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 23)
Which one of the following molecules is polar?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 24)
Bond distance in HF is \[~9.17\times {{10}^{-11}}m.\] Dipole moment of HF is \[6.104\times {{10}^{-30}}Cm.\] The percentage ionic character in HF will be: (electron charge\[=1.60\times {{10}^{-19}}C\])
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 25)
Dipole moment of\[{{H}_{2}}O\] is 1.85 D. If the bond angle is \[~105{}^\circ \] and\[O-H\] bond length is\[~0.94\overset{\text{o}}{\mathop{\text{A}}}\,\], what is the magnitude of charge on the oxygen atom in water molecule?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 26)
Which of the correct increasing order of lone pair of electrons on the central atom?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 27)
The molecules \[B{{F}_{3}}\] and \[N{{F}_{3}}\] are both covalent compounds, but \[B{{F}_{3}}\] is non polar whereas \[N{{F}_{3}}\] is polar. The reason for this is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 28)
Among the following chloro-compound having the lowest dipole moment is
A)
done
clear
B)
C)
done
clear
D)
View Solution play_arrow
question_answer 29)
Although\[~C{{N}^{-}}\] ion and \[{{N}_{2}}\] molecule are isoelectronic, yet\[{{N}_{2}}\] molecule is chemically inert because of
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 30)
The number and type of bonds in ion in\[~Ca{{C}_{2}}\]are:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 31)
Among the following compounds the one that is polar and has the central atom with \[~s{{p}^{2}}\] hybridization is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 32)
Among the following ions the \[~p\pi -d\pi \] overlap could be present in
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 33)
In the reaction \[2PC{{l}_{5}}\rightleftharpoons PCl_{_{4}}^{-}+PCl_{6}^{-}\], the change in hybridization is from
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 34)
In an octahedral structure, the pair of d orbitals involved in\[~{{d}^{2}}s{{p}^{3}}\] hybridization is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 35)
\[{{N}_{2}}\] and\[{{O}_{2}}\] are converted into monocations, \[N_{_{2}}^{+}\] and\[O_{_{2}}^{+}\] respectively. Which of the following statements is wrong?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 36)
The correct order of bond dissociation energy among \[{{N}_{2}},\,{{O}_{2}},O_{2}^{-}\] is shown in which of the following arrangements?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 37)
In\[Xe{{F}_{2}},\,\,Xe{{F}_{4}}\] and \[Xe{{F}_{6}}\,,\] the number of lone pairs on Xe are respectively
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 38)
Which of the following represents the given mode of hybridisation \[s{{p}^{2}}-s{{p}^{2}}-sp-sp\] from left to right?
A)
done
clear
B)
done
clear
C)
done
clear
D)
View Solution play_arrow
question_answer 39)
In \[Br{{F}_{3}}\] molecule, the lone pairs occupy equatorial positions to minimize
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 40)
The group of molecules having identical shape is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 41)
\[S{{F}_{2}},S{{F}_{4}}\] and \[S{{F}_{6}}\] have the hybridization at sulphur atom respectively as:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 42)
Which of the following molecules has two sigma \[(\sigma )\] and two pi\[(\pi )\] bonds?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 43)
The shape of \[IF_{6}^{-}\] is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 44)
Which of the following statement is correct?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 45)
The maximum number of \[~90{}^\circ \] angles between bond pair-bond pair of electrons is observed in
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 46)
The true statements from the following are
1. \[P{{H}_{5}}\] and \[BiC{{l}_{5}}\] do not exist 2. \[p\pi -d\pi \] bond is present in \[S{{O}_{2}}\] 3. Electrons travel with the speed of light 4. \[Se{{F}_{4}}\]and \[C{{H}_{4}}\]have same shape 5. \[I_{3}^{+}\]has bent geometry
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 47)
KF combines with HF to form \[KH{{F}_{2}}\]. The compound contains the species
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 48)
Which of the following statements is incorrect?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 49)
In which of the following sets, all the given species are isostructural?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 50)
The formation of molecular complex \[B{{F}_{3}}-N{{H}_{3}}\] results in a change in hybridization of boron
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 51)
Which of the following substances has the least ionic character?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 52)
Sulphur reacts with chlorine in 1 : 2 ratio and forms X. Hydrolysis of X gives a sulphur compound Y. What is the hybridisation state of central atom in the compound.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 53)
CaO and \[~NaCl\] have the same crystal structure and approximately the same ionic radii. If U is the lattice energy of \[~NaCl\], the approximate lattice energy of \[CaO\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 54)
Dipole moment is shown by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 55)
Which of the following has the square planar structure?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 56)
Which one of the following molecules is paramagnetic?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 57)
Which one of the following pairs of molecules will have permanent dipole moments for both members?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 58)
In which of the following ionization processes the bond energy has increased and also the magnetic behaviour has changed from paramagnetic to diamagnetic?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 59)
Bond order normally gives idea of stability of a molecular species. All the molecules viz. \[{{H}_{2}},L{{i}_{2}}\]and\[{{B}_{2}}\]have the same bond order yet they are not equally stable. Their stability order is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 60)
The internuclear distances in \[~O-O\] bonds for \[O_{2}^{+},{{O}_{2}},O_{2}^{-}\] and \[O_{2}^{2-}\] respectively are:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
![]() the hybrid state assumed by N is
the hybrid state assumed by N is  done
clear
done
clear
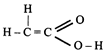 done
clear
done
clear
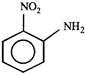 done
clear
done
clear
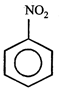 done
clear
done
clear
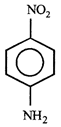 done
clear
done
clear
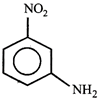 done
clear
done
clear
