-
question_answer1)
The molality of a urea solution in which 0.0200 g of urea (\[N{{H}_{2}}CON{{H}_{2}}\]) is added to 0.400 \[d{{m}^{3}}\] of water at STP is
A)
\[0.555\,mol\,k{{g}^{-1}}\] done
clear
B)
\[5.55\times {{10}^{-4}}\,mol\,k{{g}^{-1}}\] done
clear
C)
\[8.33\times {{10}^{-4}}\,mol\,k{{g}^{-1}}\] done
clear
D)
\[33.3\,mol\,k{{g}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer2)
Molar solubility of helium, nitrogen and oxygen are plotted against partial pressure of the gas at constant temperature. Henry's law constant for hese gases will lie in the following sequence?
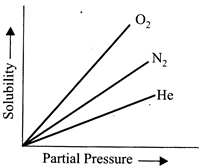
A)
\[{{O}_{2}}>{{N}_{2}}>He\] done
clear
B)
\[{{O}_{2}}<{{N}_{2}}<He\] done
clear
C)
\[{{O}_{2}}={{N}_{2}}=He\] done
clear
D)
\[{{O}_{2}}>{{N}_{2}}>He\] done
clear
View Solution play_arrow
-
question_answer3)
Henry's law constant for \[{{N}_{2}}\] at 310 K is 82.35 kbar. \[{{N}_{2}}\] exerts a partial pressure of 0.840 bar. If \[{{N}_{2}}\] gas is bubbled through water at 293 K, then the number of millimoles of \[{{N}_{2}}\] that will dissolve in 1 L of water is
A)
\[0.0716\] done
clear
B)
\[1.30\times {{10}^{-5}}\] done
clear
C)
\[1.25\times {{10}^{-2}}\] done
clear
D)
0.0555 done
clear
View Solution play_arrow
-
question_answer4)
Out of the following liquid pair which solution follows the positive deviation from Raoult's law?
A)
Acetone + Chloroform done
clear
B)
Water + Nitric acid done
clear
C)
Water + Hydrochloric acid done
clear
D)
Benzene + Methanol done
clear
View Solution play_arrow
-
question_answer5)
What mass of non-volatile solute (urea) needs to be dissolved in 150 g of water in order to decrease the vapour pressure of water by 35%. What will be the molality of solution?
A)
23.88 done
clear
B)
31.12 done
clear
C)
25.47 done
clear
D)
29.92 done
clear
View Solution play_arrow
-
question_answer6)
An ideal mixture of liquids A and B with 2 moles of A and 2 moles of B has a total vapour pressure of 1 atm at a certain temperature. Another mixture with 1 mole of A and 3 moles of B has a vapour pressure greater than 1 atm. But if 4 moles of C are added to the second mixture, the vapour pressure comes down to 1 atm. Vapour pressure of C, \[P_{c}^{0}\]=0.8 atm. Calculate the vapour pressures of pure A and pure B
A)
\[P_{A}^{o}\]=1.4 atm, \[P_{B}^{o}\]=0.7 atm done
clear
B)
\[P_{A}^{o}\]=1.2atm, \[P_{B}^{o}\]=0.6atm done
clear
C)
\[P_{A}^{o}\]=1.4atm, \[P_{B}^{o}\]=0.6atm done
clear
D)
\[P_{A}^{o}\]= 0.6 atm, \[P_{B}^{o}\]= 1.4 atm done
clear
View Solution play_arrow
-
question_answer7)
The vapour pressure of n-hexane at 350 K is 840 torr and that of cyclohexane is 600 torr. Mote fraction of hexane in the mixture that boils at 350 K and 1 atm pressure assuming ideal behaviour is
A)
0.67 done
clear
B)
0.80 done
clear
C)
0.50 done
clear
D)
2.2 done
clear
View Solution play_arrow
-
question_answer8)
A current of dry air is passed through a bulb containing 5 g of a solute in 100 g of water and then through water alone. The losses in weight of the solution and pure water were respectively 0.78 g and 0.02 g. Calculate relative lowering of vapour pressure.
A)
2.04 done
clear
B)
1.05 done
clear
C)
0.03 done
clear
D)
0.09 done
clear
View Solution play_arrow
-
question_answer9)
How many grams of sucrose (M. wt. = 342) should be dissolved in 100 g water in order to produce a solution with a \[105.0{}^\circ C\] difference between the boiling point and the freezing temperatures? \[({{K}_{f}}=\text{ }1.86\text{ }C\text{/}m,\,\,{{K}_{b}}=0.51{}^\circ C\text{/}m)\]
A)
34.2 g done
clear
B)
72 g done
clear
C)
342 g done
clear
D)
460 g done
clear
View Solution play_arrow
-
question_answer10)
Which of the given aqueous solution has the maximum boiling point? \[({{K}_{b}}\,of\,{{H}_{2}}O)=0.52{}^\circ \,mo{{l}^{-1}}kg),\,{{T}_{b}}({{H}_{2}}O)=373K)\]
A)
6% urea solution by weight of solution done
clear
B)
18% glucose solution by weight of solution done
clear
C)
34.2% sucrose solution by weight of solution done
clear
D)
All have equal boiling points done
clear
View Solution play_arrow
-
question_answer11)
The boiling point elevation constant for benzene is \[2.57{}^\circ C/m.\] The boiling point of benzene is \[81{}^\circ C\]. Determine the boiling point of solution formed when 10 g of \[{{C}_{4}}{{H}_{12}}\]is dissolved in 20 g benzene.
A)
71.46 done
clear
B)
7.14 done
clear
C)
85.76 done
clear
D)
88.14 done
clear
View Solution play_arrow
-
question_answer12)
The cryoscopic constant for acetic acid is 3.6 K kg/mol. A solution of 1 g of a hydrocarbon in 100 g of acetic acid freezes at \[16.14{}^\circ C\]instead of the usual\[16.60{}^\circ C\]. The hydrocarbon contains 92.3% carbon. What is the'molecular formula?
A)
\[{{C}_{6}}{{H}_{6}}\] done
clear
B)
\[{{C}_{6}}{{H}_{12}}\] done
clear
C)
\[{{C}_{8}}{{H}_{18}}\] done
clear
D)
\[{{C}_{10}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer13)
Solute A is a ternary electrolyte and solute B is non-electrolyte. If 0.1 M solution of solute B produces an osmotic pressure of 2P, then 0.05 M solution of A at the same temperature will produce an osmotic pressure equal to:
A)
P done
clear
B)
1.5P done
clear
C)
IP done
clear
D)
3P done
clear
View Solution play_arrow
-
question_answer14)
Freezing point of a biological fluid is \[-0.60{}^\circ C\]in aqueous solution \[{{K}_{f}}({{H}_{2}}O)=\text{ }1.86{}^\circ mo{{l}^{-}}~kg.\] Thus, its osmotic pressure at 310 K is (assume molarity = molality)
A)
0.0766 atm done
clear
B)
7.66 atm done
clear
C)
0.766 atm done
clear
D)
8.19 atm done
clear
View Solution play_arrow
-
question_answer15)
Phenol associates in benzene to a certain extent to form a dimer. A solution containing \[20\times {{10}^{-3}}\] kg of phenol in 1.0 kg-of benzene has its freezing point depressed by 0.69 K. Calculate the fraction of phenol that has dimerised. (\[{{K}_{f}}\]for benzene = 5.12 K kg \[mo{{l}^{-1}}\])
A)
28.5% done
clear
B)
32.6% done
clear
C)
73.3% done
clear
D)
82% done
clear
View Solution play_arrow
-
question_answer16)
A living cell contains a solution which is isotonic with 0.3 (M) sugar solution. What osmotic pressure develops when the cell is placed in 0.1 (M) KCl solution at body temperature?
A)
5.08 atm done
clear
B)
2.54 atm done
clear
C)
4.92 atm done
clear
D)
2.46 atm done
clear
View Solution play_arrow
-
question_answer17)
The degree of dissociation of \[Ca{{(N{{O}_{3}})}_{2}}\] in a dilute solution containing 14 g of the salt per 200 g of water at \[100{}^\circ C\]is 70%. If the vapour pressure of water is 760 mmHg, calculate the vapour pressure of solution.
A)
750.6 mmHg done
clear
B)
755.8 mmHg done
clear
C)
745.98 mmHg done
clear
D)
739.56 mmHg done
clear
View Solution play_arrow
-
question_answer18)
In a mixture of A and B, components show -ve deviations as,
A)
\[\Delta {{V}_{mix}}\,\text{is}+ve\] done
clear
B)
\[A-B\] interaction is weaker than \[A-A\] and \[B-B\] interaction done
clear
C)
\[\Delta {{H}_{mix}}\,\text{is}+ve\] done
clear
D)
A-B interaction is stronger than \[A-A\] and \[B-B\] interaction done
clear
View Solution play_arrow
-
question_answer19)
The elevation in boiling point of a solution of 13.44 g of\[CuC{{l}_{2}}\], (molecular weight = 134.4, \[{{K}_{b}}\] = 0.52 K \[molalit{{y}^{-1}}\] in 1 kg water using the given information will be
A)
0.16 done
clear
B)
0.05 done
clear
C)
0.1 done
clear
D)
0.2 done
clear
View Solution play_arrow
-
question_answer20)
The degree of dissociation (1) of a weak electrolyte \[{{A}_{x}}{{B}_{y}}\] is related to van't Hoff factor (i) by the expression
A)
\[\alpha =\frac{i-1}{(x+y-1)}\] done
clear
B)
\[\alpha =\frac{i-1}{x+y+1}\] done
clear
C)
\[\alpha =\frac{x+y-1}{i-1}\] done
clear
D)
\[\alpha =\frac{x+y+1}{i-1}\] done
clear
View Solution play_arrow
-
question_answer21)
The mass of a non-volatile solute of molar mass 40 g \[mo{{l}^{-1}}\], which on dissolution in 57 g octane to reduce the vapom pressure of octane by 20%, is
View Solution play_arrow
-
question_answer22)
A 1.17% solution of NaCl is isotonic with 7.2% solution of glucose. Calculate the van't Hoff factor of NaCl.
View Solution play_arrow
-
question_answer23)
A mixture of two immiscible liquids nitrobenzene and water boiling at \[99{}^\circ C\] has a partial pressure of watd 733 mm and of nitrobenzene 27 mm. The ratio of mass of water and nitrobenzene in mixture is....
View Solution play_arrow
-
question_answer24)
An aqueous solution of a substance with molar mass 240 has osmotic pressure 0.2 atm at 300 K. The density solution in \[g\text{/}d{{m}^{3}}\] is: (R = 0.08 litre atm \[{{K}^{-1}}\]\[mo{{l}^{-1}}\])
View Solution play_arrow
-
question_answer25)
A solution of 6.2 g of ethylene glycol in 55 g \[{{H}_{2}}O\] is cooled to \[-\,3.72{}^\circ C\]. The ice separated from the solution is: (\[{{K}_{f}}\]of \[{{H}_{2}}O\]= 1.86 K\[molalit{{y}^{-1}}\])
View Solution play_arrow
