-
question_answer1)
Among the following the state function(s) is (are)
| (I) Internal energy |
| (II) Irreversible expansion work |
| (III) Reversible expansion work |
| (IV) Molar enthalpy |
A)
(ii) and (iii) done
clear
B)
(i), (ii) and (iii) done
clear
C)
(i) and (iv) done
clear
D)
(i) only done
clear
View Solution play_arrow
-
question_answer2)
If a reaction involves only solids and liquids which of the following is true?
A)
\[\Delta H<\Delta E\] done
clear
B)
\[\Delta H=\Delta E\] done
clear
C)
\[\Delta H>\Delta E\] done
clear
D)
\[\Delta H=\Delta E+\Delta nRT\] done
clear
View Solution play_arrow
-
question_answer3)
One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0 L, 95 K) \[\to \] (4.0 atm, 5.0 L, 245 K) with a change in internal energy, \[\Delta U=30.0\text{ }L\]atm. The change in enthalpy \[\Delta H\]of the process in L atm is.
A)
40.0 done
clear
B)
42.3 done
clear
C)
44.0 done
clear
D)
Not defined because pressure is not constant done
clear
View Solution play_arrow
-
question_answer4)
If bond enthalpies of \[N\equiv N,\text{ }H-H\] and \[N-H\] bonds are \[{{x}_{1}},{{x}_{2}}\] and \[{{x}_{3}}\] respectively, \[\Delta H_{f}^{o}\] for \[N{{H}_{3}}\] will be
A)
\[{{x}_{1}}+3{{x}_{2}}-6{{x}_{3}}\] done
clear
B)
\[1/2{{x}_{1}}+3/2{{x}_{2}}-3{{x}_{3}}\] done
clear
C)
\[3{{x}_{3}}-1/2{{x}_{1}}-3/2{{x}_{2}}\] done
clear
D)
\[6{{x}_{3}}-{{x}_{1}}-3{{x}_{2}}\] done
clear
View Solution play_arrow
-
question_answer5)
Heat of neutralization of a strong acid HA and a weaker acid HB with KOH are \[-13.7\] and\[-12.7\text{ }k\text{ }cal\text{ }mo{{l}^{-1}}\]. When 1 mole of KOH was added to a mixture containing 1 mole each of HA and HB, the heat change was \[-13.5\] kcal. In what ratio is the base distributed between HA and HB.
A)
3 : 1 done
clear
B)
1 : 3 done
clear
C)
4 : 1 done
clear
D)
1 : 4 done
clear
View Solution play_arrow
-
question_answer6)
A gas expands adiabatically at constant pressure such that \[T\propto {{V}^{-1/2}}\]. The value of \[\gamma ({{C}_{p,m}}/{{C}_{V,m}})\]of the gas will be:
A)
1.30 done
clear
B)
1.50 done
clear
C)
1.70 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer7)
One mole of an ideal gas at 300 K is expanded isothermally from an initial volume of 1 litre to 10 litres. The AE for this process is \[(R=2\text{ }cal\text{ }mo{{l}^{-1}}{{k}^{-1}})\]
A)
163.7 cal done
clear
B)
zero done
clear
C)
1381.1 cal done
clear
D)
9 lit atm done
clear
View Solution play_arrow
-
question_answer8)
The latent heat of vapourization of \[\varepsilon \] liquid at 500 K and 1 atm pressure is 10.0 kcal/mol. What will be the change in internal energy \[\left( \Delta U \right)\] of 3 moles of liquid at the same temperature
A)
\[13.0\text{ }kcal/mol\] done
clear
B)
\[-13.0\text{ }kcal/mol\] done
clear
C)
\[27.0\text{ }kcal\] done
clear
D)
\[-7.0\text{ }kcal/mol\] done
clear
View Solution play_arrow
-
question_answer9)
Equal volumes of methanoic acid and sodium hydroxide are mixed. If x is the heat of formation of water, then heat evolved on neutralization is
A)
more than x done
clear
B)
equal to x done
clear
C)
less than x done
clear
D)
twice x. done
clear
View Solution play_arrow
-
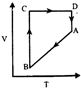
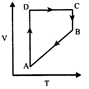
question_answer10)
A cyclic process ABCD is shown in P-V diagram for an ideal gas. Which of the following diagram represents the same process?
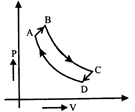
A)
B)
C)
D)
View Solution play_arrow
-
question_answer11)
What is the value of change in internal energy at 1 atm in the process?
| \[{{H}_{2}}O\left( 1,323\text{ }K \right)\xrightarrow{{}}{{H}_{2}}O\left( g,423K \right)\] |
| Given: \[{{C}_{v,m}}({{H}_{2}}O,I)=75.0J{{K}^{-1}}mo{{l}^{-1}}\]; |
| \[{{C}_{p.m}}({{H}_{2}}O,g)=33.314J{{K}^{-1}}mo{{l}^{-1}}\] |
| \[\Delta {{H}_{vap}}\,\text{at}\,373\,K=40.7\,kJ\text{/}mol\] |
A)
\[42.91\text{ }kJ/mol\] done
clear
B)
\[43086\text{ }kJ/mol\] done
clear
C)
\[42.6\text{ }kJ/mol\] done
clear
D)
\[49.6\text{ }kJ/mol\] done
clear
View Solution play_arrow
-
question_answer12)
Which of the following statements/relationships is not correct in thermodynamic changes?
A)
\[\Delta U=0\] (isothermal reversible expansion of a gas) done
clear
B)
\[w=-nRT\] In \[\frac{{{V}_{2}}}{{{V}_{1}}}\] (isothermal reversible expansion of an ideal gas) done
clear
C)
\[w=nRT\] In \[\frac{{{V}_{2}}}{{{V}_{1}}}\] (isothermal reversible expansion of an ideal gas) done
clear
D)
For a system of constant volume heat involved directly changes to internal energy. done
clear
View Solution play_arrow
-
question_answer13)
The heat of atomization of methane and ethane are \[360\text{ }kJ/mol\] and\[620\text{ }kJ/mol\], respectively. The longest wavelength of light capable of breaking the \[C-C\] bond is (Avogadro number \[=6.02\times {{10}^{23}}mo{{l}^{-1}},\,\,h=6.62\times {{10}^{-34}}Js):\]
A)
\[2.48\times {{10}^{4}}nm\] done
clear
B)
\[1.49\times {{10}^{3}}nm\] done
clear
C)
\[2.48\times {{10}^{3}}nm\] done
clear
D)
\[1.49\times {{10}^{4}}nm\] done
clear
View Solution play_arrow
-
question_answer14)
Following reaction occurrs in an automobile \[2{{C}_{8}}{{H}_{18}}\left( g \right)+25{{O}_{2}}\left( g \right)\to 16C{{O}_{2}}\left( g \right)+18{{H}_{2}}O\left( g \right).\]The sign of \[\Delta H,\Delta S\] and AG would be
A)
\[+,-,+\] done
clear
B)
\[-,+,-\] done
clear
C)
\[-,+,+\] done
clear
D)
\[+,+,-\] done
clear
View Solution play_arrow
-
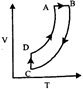
question_answer15)
A heat engine carries one mole of an ideal mono-atomic gas around the cycle as shown in the figure. Select the correct option:
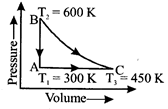
A)
\[{{q}_{AB}}=450R\] and \[{{q}_{CA}}=-450R\] done
clear
B)
\[{{q}_{AB}}=450R\] and \[{{q}_{CA}}=-225R\] done
clear
C)
\[{{q}_{AB}}=450R\] and \[{{q}_{CA}}=-375R\] done
clear
D)
\[{{q}_{AB}}=375R\] and \[{{q}_{CA}}=-450R\] done
clear
View Solution play_arrow
-
question_answer16)
The standard enthalpy of formation of \[N{{H}_{3}}\]is\[-46.0\text{ }kJ/mol\]. If the enthalpy of formation of \[{{H}_{2}}\] from its atoms is \[-436\text{ }kJ/mol\] and that of \[{{N}_{2}}\] is\[-712\text{ }kJ/mol\], the average bond enthalpy of \[N-H\] bonding is:
A)
\[-1102\text{ }kJ/mol\] done
clear
B)
\[-964\text{ }kJ/mol\] done
clear
C)
\[+352\text{ }kJ/mol\] done
clear
D)
\[+1056\text{ }kJ/mol\] done
clear
View Solution play_arrow
-
question_answer17)
The standard enthalpy of formation \[({{\Delta }_{f}}{{H}^{o}}_{298})\] for methane, \[C{{H}_{4}}\] is\[-74.9\text{ }kJ\text{ }mo{{l}^{-1}}\]. In order to calculate the average energy given out in the formation of a \[C-H\] bond from this it is necessary to know which one of the following?
A)
The dissociation energy of the hydrogen molecule, \[{{H}_{2}}\]. done
clear
B)
The first four ionisation energies of carbon. done
clear
C)
The dissociation energy of \[{{H}_{2}}\] and enthalpy of sublimation of carbon (graphite). done
clear
D)
The first four ionisation energies of carbon and electron affinity of hydrogen. done
clear
View Solution play_arrow
-
question_answer18)
Assuming that water vapour is an ideal gas, the internal energy change \[\left( \Delta U \right)\] when 1 mol of water is vapourised at 1 bar pressure and \[100{}^\circ C,\] (given : molar enthalpy of vapourisation of water at 1 bar and \[373\text{ }K=41\text{ }kJ\text{ }mo{{l}^{-1}}\] and \[R=8.3\,J\,mo{{l}^{-1}}{{K}^{-1}}\]) will be
A)
\[41.00kJ\,mo{{l}^{-1}}\] done
clear
B)
\[4.100kJ\,mo{{l}^{-1}}\] done
clear
C)
\[3.7904\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
D)
\[37.904\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer19)
An ideal gas expands against a constant external pressure of 2.0 atmosphere from 20 litre to 40 litre and absorbs 10 kJ of heat from surrounding. What is the change in internal energy of the system? (given : 1 atm-litre = 101.3 J)
A)
4052 J done
clear
B)
5948 J done
clear
C)
14052 J done
clear
D)
9940 J done
clear
View Solution play_arrow
-
question_answer20)
For which one of the processes represented by the following equations the enthalpy (heat) change is likely to be negative
A)
\[C{{l}^{-}}(g)+aq\to C{{l}^{-}}(aq)\] done
clear
B)
\[Cl(g)\to C{{l}^{+}}(g)+{{e}^{-}}\] done
clear
C)
\[1/2C{{l}_{2}}(g)\to Cl(g)\] done
clear
D)
\[C{{l}_{2}}(l)\to C{{l}_{2}}(g)\] done
clear
View Solution play_arrow
-
question_answer21)
The "thermite reaction' involving the reaction between ferric oxide and metallic aluminium produces molten iron. Given that \[2A1+3/2{{O}_{2}}\to A{{l}_{2}}{{O}_{3}};\Delta {{H}_{1}}=-400\,kcal/mol\] \[2Fe+3/2{{O}_{2}}\to F{{e}_{2}}{{O}_{3}};\Delta {{H}_{2}}=-200\,kcal/mol.\] What is \[\Delta H\] for the formation of 1 mole of iron?
A)
\[-100kcal\] done
clear
B)
\[-200\text{ }kcal\] done
clear
C)
\[+100\text{ }kcal\] done
clear
D)
\[+200\text{ }kcal\] done
clear
View Solution play_arrow
-
question_answer22)
Given:
| (I) \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\to {{H}_{2}}O(l);\]\[\Delta {{H}^{o}}_{298k}=-285.9\,kJ\,mo{{l}^{-1}}\] |
| (II) \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\to {{H}_{2}}O(g);\]\[\Delta {{H}^{o}}_{298k}=-241.8\,kJ\,mo{{l}^{-1}}\] |
| The molar enthalpy of vapourisation of water will be: |
A)
\[241.8\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[~22.0\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[44.1kJ\,mo{{l}^{-1}}\] done
clear
D)
\[527.7\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer23)
Given
| Reaction | Energy Change (in KJ) |
| \[Li\left( s \right)\to Li\left( g \right)\] | 161 |
| \[Li\left( g \right)\to L{{i}^{+}}\left( g \right)\] | 520 |
| \[\frac{1}{2}{{F}_{2}}(g)\to F(g)\] | 77 |
| \[F\left( g \right)+{{e}^{-}}\to {{F}^{-}}\left( g \right)\] | Electron gain enthalpy |
| \[L{{i}^{+}}(g)+{{F}^{-}}(g)\to LiF(s)\] | -1047 |
| \[Li(s)+\frac{1}{2}{{F}_{2}}(g)\to LiF(s)\] | -617 |
Based on data provided, the value of electron gain enthalpy of fluorine would be:
A)
\[-300\,kJ\,mo{{l}^{-1}}\] done
clear
B)
\[-350kJ\,mo{{l}^{-1}}\] done
clear
C)
\[-328\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
D)
\[-228\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer24)
Fixed mass of an ideal gas contained in a 24.63 L sealed rigid vessel at 1 atm is heated from -\[73{}^\circ C\] to\[27{}^\circ C\]. Calculate change in gibb's energy if entropy of gas is a function of temperature as \[S=2+{{10}^{-2}}T\text{ }\left( J/K \right):\](Use 1 atm\[L=0.1\text{ }kJ\])
A)
1231.5 J done
clear
B)
1281.5 J done
clear
C)
781.5J done
clear
D)
0 done
clear
View Solution play_arrow
-
question_answer25)
For the reaction taking place at certain temperature \[N{{H}_{2}}COON{{H}_{4}}(s)\rightleftharpoons 2N{{H}_{3}}(g)+C{{O}_{2}}(g),\] if equilibrium pressure is 3X bar then \[{{\Delta }_{\text{r}}}G{}^\circ \] would be
A)
\[-RT\] ln \[9-3RT\] ln X done
clear
B)
RT ln \[4-3RT\] ln X done
clear
C)
\[-3RT\] ln X done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer26)
Standard Gibb's free energy change for isomerization reaction cis-2-pentene \[\rightleftharpoons \] trans-2-pentene is \[-3.67\text{ }kJ/mol\] at 400 K. If more trans-2-pentene is added to the reaction vessel, then
A)
more cis-2-pentene is formed done
clear
B)
equilibrium remains unaffected done
clear
C)
additional trans-2-pentene is formed done
clear
D)
equilibrium is shifted in forward direction done
clear
View Solution play_arrow
-
question_answer27)
The dissolution of \[KCl\] in water is endothermic yet it dissolves in water spontaneously. Which one of the following best explains this behaviour?
A)
Endothermic processes are energetically favoured. done
clear
B)
The electrostatic bonds between the ions are not too strong. done
clear
C)
Energy changes have nothing to do with the dissolution processes. done
clear
D)
The entropy driving force causes the dissolution. done
clear
View Solution play_arrow
-
question_answer28)
Hydrogen has an ionisation energy of \[1311\text{ }kJ\text{ }mo{{l}^{-1}}\] and for chlorine it is \[1256\text{ }kJ\text{ }mo{{l}^{-1}}.\] Hydrogen forms \[{{H}^{+}}\left( aq \right)\] ions but chlorine does not form \[C{{l}^{+}}(aq)\] ions because
A)
\[{{H}^{+}}\] has lower hydration enthalpy done
clear
B)
\[C{{l}^{+}}\]has lower hydration enthalpy done
clear
C)
\[Cl\] has high electron affinity done
clear
D)
\[Cl\] has high electronegativity done
clear
View Solution play_arrow
-
question_answer29)
At \[25{}^\circ C\], when 1 mole of \[MgS{{O}_{4}}\] was dissolved in water, the heat evolved was found to be \[91.2\text{ }kJ.\] One mole of \[MgS{{O}_{4}}\]. \[7{{H}_{2}}O\] on dissolution gives a solution of the same composition accompanied by an absorption of \[13.8\text{ }kJ\]. The enthalpy of hydration, i.e., \[\Delta {{H}_{h}}\] for the reaction \[MgS{{O}_{4}}\left( s \right)+7{{H}_{2}}O\left( l \right)\xrightarrow{{}}MgS{{l}_{4}}.7{{H}_{2}}O\left( s \right)\] is:
A)
\[-105\text{ }kJ/mol\] done
clear
B)
\[-77.4\text{ }kJ/mol\] done
clear
C)
\[105\text{ }kJ/mol\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer30)
Calculate the heat produced (in kJ) when 224 g of \[CaO\] is completely converted to \[CaC{{O}_{3}}\]by reaction with \[C{{O}_{2}}\] at \[27{}^\circ C\] in a container of fixed volume.
| Given: \[\Delta H_{f}^{o}\left( CaC{{O}_{3}},s \right)=-1207\text{ }kJ/mol;\] |
| \[\Delta H_{f}^{o}(CaO,s)=-\,635\,kJ/mol,\] |
| \[\Delta H_{f}^{o}(C{{O}_{2}},g)=-\text{ }394\text{ }kJ\text{/}mol;\] [Use\[R=8.3\text{ }J{{K}^{-1}}mo{{l}^{-1}}\]] |
A)
\[702.04\text{ }kJ\] done
clear
B)
\[721.96\,kJ\] done
clear
C)
\[712\text{ }kJ\] done
clear
D)
\[721\text{ }kJ\] done
clear
View Solution play_arrow
-
question_answer31)
Given that
| (i) \[{{\Delta }_{f}}H{}^\circ \] of \[{{N}_{2}}O\] is \[82\text{ }kJ\text{ }mo{{l}^{-1}}\] |
| (ii) Bond energies of \[N\equiv N,N=N,O=O\] and \[N=O\] are 946, 418, 498 and \[607kJ\text{ }mo{{l}^{-1}}\] respectively, The resonance energy of \[{{N}_{2}}O\] is: |
A)
\[-88kJ\] done
clear
B)
\[-66kJ\] done
clear
C)
\[-62kJ\] done
clear
D)
\[~-\,44\,kJ\] done
clear
View Solution play_arrow
-
question_answer32)
The difference between the reaction enthalpy change \[({{\Delta }_{r}}H)\] and reaction internal energy change \[({{\Delta }_{r}}U)\] for the reaction: \[2{{C}_{6}}{{H}_{6}}\left( l \right)+15{{O}_{2}}\left( g \right)\xrightarrow{{}}12C{{O}_{2}}\left( g \right)+6{{H}_{2}}O\left( l \right)\]at \[300K\] is \[(R=8.314\,Jmo{{l}^{-1}}{{K}^{-1}})\]
A)
\[0J\,mo{{l}^{-1}}\] done
clear
B)
\[2490\text{ }J\text{ }mo{{l}^{-1}}\] done
clear
C)
\[-2490J\text{ }mo{{l}^{-1}}\] done
clear
D)
\[-7482J\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer33)
From given following equations and \[\Delta H{}^\circ \] values, determine the enthalpy of reaction at 298 K for the reaction:
| \[{{C}_{2}}{{H}_{4}}\left( g \right)+6{{F}_{2}}\left( g \right)\xrightarrow{{}}2C{{F}_{4}}\left( g \right)+4HF\left( g \right)\] |
| \[{{H}_{2}}(g)+{{F}_{2}}(g)\xrightarrow{{}}2HF(g);\Delta H_{1}^{{}^\circ }=-537kJ\] |
| \[C\left( s \right)+2{{F}_{2}}\left( g \right)\xrightarrow{{}}C{{F}_{4}}\left( g \right);\text{ }\Delta H_{2}^{{}^\circ }=-680\text{ }kJ\] |
| \[2C\left( s \right)+2{{H}_{2}}\left( g \right)\xrightarrow{{}}{{C}_{2}}{{H}_{4}}\left( g \right);\Delta H_{3}^{{}^\circ }=52kJ\] |
A)
\[~-1165\] done
clear
B)
\[-2486\] done
clear
C)
\[+1165\] done
clear
D)
\[+2486\] done
clear
View Solution play_arrow
-
question_answer34)
Substance \[{{A}_{2}}B(g)\] can undergoes decomposition to form two set of products:
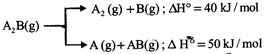
If the molar ratio of \[{{A}_{2}}(g)\] to A (g) is 5 : 3 in a set of product gases, then the energy involved in the decomposition of 1 mole of \[{{A}_{2}}B(g)\] is:
A)
\[48.75kJ/mol\] done
clear
B)
\[43.73\text{ }kJ/mol\] done
clear
C)
\[46.25\text{ }kJ/mol\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer35)
\[36mL\] of pure water takes 100 sec to evaporate from a vessel and heater connected to an electric source which delivers 806 watt. The\[\Delta {{H}_{vap}}\]of \[{{H}_{2}}O\] is:
A)
\[40.3\text{ }kJ/mol\] done
clear
B)
\[43.2\text{ }kJ/mol\] done
clear
C)
\[4.03\text{ }kJ/mol\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer36)
For the combustion reaction at \[298\text{ }K\] \[2Ag\left( s \right)+1/2{{O}_{2}}\left( g \right)\xrightarrow{{}}2A{{g}_{2}}O\left( s \right)\] Which of the following alternatives is correct?
A)
\[\Delta H=\Delta U\] done
clear
B)
\[\Delta H>\Delta U\] done
clear
C)
\[\Delta H<\Delta U\] done
clear
D)
\[\Delta H\] and \[\Delta U\] has no relation with each other done
clear
View Solution play_arrow
-
question_answer37)
A gas undergoes change from state A to state B. In this process, the heat absorbed and work done by the gas is 5 J and 8 J, respectively. Now gas is brought back to A by another process during which 3 J of heat is evolved. In this reverse process of B to A:
A)
\[10\text{ }J\] of the work will be done by the gas. done
clear
B)
\[6\text{ }J\] of the work will be done by the gas. done
clear
C)
\[10\text{ }J\] of the work will be done by the surrounding on gas. done
clear
D)
\[6\text{ }J\] of the work will be done by the surrounding on gas. done
clear
View Solution play_arrow
-
question_answer38)
The amount of energy released when \[20mL\] of \[0.5M\text{ }NaOH\] are mixed with \[100\text{ }mL\] of 0.1 M \[HCl\] is \[x\text{ }kJ.\] The heat of neutralisation (in kJ\[mo{{l}^{-1}}\]) is
A)
\[-100x\] done
clear
B)
\[-50x\] done
clear
C)
\[+100x\] done
clear
D)
\[+50x\] done
clear
View Solution play_arrow
-
question_answer39)
Which of the following value of \[{{\Delta }_{f}}{{H}^{{}^\circ }}\] represent that the product is least stable?
A)
\[-94.0\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-231.6\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
C)
\[+21.4\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
D)
\[+64.8\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer40)
Enthalpy of neutralisation of \[HCl\] with \[NaOH\] is x. The heat evolved when \[500\text{ }mL\] of \[2\text{ }N\text{ }HCl\] are mixed with \[250\text{ }mL\] of \[4N\text{ }NaOH\] will be.
A)
\[500x\] done
clear
B)
\[100x\] done
clear
C)
\[x\] done
clear
D)
\[~10x\] done
clear
View Solution play_arrow
-
question_answer41)
The enthalpy of neutralisation of \[N{{H}_{4}}OH\] with \[HCl\] is \[-51.46\text{ }kJ\text{ }mo{{l}^{-1}}\] and the enthalpy of neutralization of \[NaOH\] with \[HCl\] is \[-55.90\text{ }kJ\text{ }mo{{l}^{-1}}\]. The enthalpy of ionisation of \[N{{H}_{4}}OH\] is
A)
\[-107.36 kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-4.44 kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[+107.36\,kJ\,mo{{l}^{-1}}\] done
clear
D)
\[+4.44\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer42)
The molar heat capacity \[\left( {{C}_{p}} \right)\] of \[C{{D}_{2}}O\] is 10 cals at 1000 K. The change in entropy associated with cooling of 32 g of \[C{{D}_{2}}O\] vapour from 1000 K to 100 K at constant pressure will be: (D = deuterium, atomic mass = 2 u)
A)
\[23.03\text{ }cal\text{ }de{{g}^{-1}}\] done
clear
B)
\[-23.03\text{ }cal\text{ }de{{g}^{-1}}\] done
clear
C)
\[2.303\text{ }cal\text{ }de{{g}^{-1}}\] done
clear
D)
\[-2.303\text{ }cal\text{ }de{{g}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer43)
The \[\left( S{}^\circ \right)\] of the following substances are:
| \[C{{H}_{4}}\left( g \right)\text{ }186.2\text{ }J{{K}^{-1}}mo{{l}^{-1}}\] |
| \[{{O}_{2}}\left( g \right)205.2\text{ }J{{K}^{-1}}mo{{l}^{-1}}\] |
| \[C{{O}_{2}}\left( g \right)213.6J{{K}^{-1}}mo{{l}^{-1}}\] |
| \[{{H}_{2}}O\left( g \right)69.9.J{{K}^{-1}}mo{{l}^{-1}}\] |
| The entropy change \[\left( \Delta {{S}^{{}^\circ }} \right)\] for the reaction |
| \[C{{H}_{4}}(g)+2{{O}_{2}}(g)\to C{{O}_{2}}(g)+2{{H}_{2}}O(l)\]is: |
A)
\[-312.5\text{ }J\,{{K}^{-1}}mo{{l}^{-1}}\] done
clear
B)
\[-242.8\text{ }J\,{{K}^{-1}}\,mo{{l}^{-1}}\] done
clear
C)
\[-108.1\text{ }J\,{{K}^{-1}}\,mo{{l}^{-1}}\] done
clear
D)
\[-37.6\text{ }J\text{ }{{K}^{-1}}\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer44)
The enthalpy of the reaction forming \[PbO\] according to the following equation is \[438\text{ }kJ\]. What heat energy (kJ) is released in formation of \[22.3g\,PbO\left( s \right)\] ? (Atomic masses: \[Pb=207,\text{ O}=16.0\]) \[2Pb\left( s \right)+{{O}_{2}}\left( g \right)\xrightarrow{{}}2PbO\left( s \right)\]
A)
21.9 done
clear
B)
28.7 done
clear
C)
14.6 done
clear
D)
34.2 done
clear
View Solution play_arrow
-
question_answer45)
The species which by definition has zero standard molar enthalpy of formation at \[298K\] is
A)
\[B{{r}_{2}}(g)\] done
clear
B)
\[C{{l}_{2}}(g)\] done
clear
C)
\[{{H}_{2}}O\left( g \right)\] done
clear
D)
\[C{{H}_{4}}(g)\] done
clear
View Solution play_arrow
-
question_answer46)
What is \[\Delta {{n}_{gas}}\] for the combustion of 1 mole of benzene, when both the reactants and the products are at 298 K ?
A)
0 done
clear
B)
1/2 done
clear
C)
3/2 done
clear
D)
\[-3/2\] done
clear
View Solution play_arrow
-
question_answer47)
The enthalpy of neutralisation of \[N{{H}_{4}}OH\]and \[C{{H}_{3}}COOH\] is \[-10.5\text{ }kcal\text{ }mo{{l}^{-1}}\] and enthalpy of neutralisation of \[C{{H}_{3}}COOH\] with strong base is\[-12.5\,kcal\text{ }mo{{l}^{-1}}\]. The enthalpy of ionisation of \[N{{H}_{4}}OH\] will be
A)
\[3.2\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
B)
\[2.0\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
C)
\[3.0\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
D)
\[4.0\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
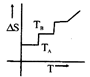
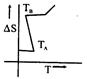
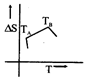
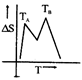
question_answer48)
If for a given substance melting point is \[{{T}_{B}}\]and freezing point is \[{{T}_{A}}\], then correct variation shown by, graph between entropy change and temperature is
A)
B)
C)
D)
View Solution play_arrow
-
question_answer49)
What is the free energy change for the conversion of 1 mole of water into steam at \[373.2\text{ }K\]. The heat of vaporization \[\left( \Delta \,{{H}_{v}} \right)\] of water of \[373.2\text{ }K\] is \[9.1\text{ }kcal\text{ }mo{{l}^{-1}}\]. The entropy change is 25.5 cal/mol deg.
A)
\[-401.6\text{ }cal/mol\] done
clear
B)
\[-416.6\text{ }cal/mol\] done
clear
C)
\[516.5\text{ }cal/mol\] done
clear
D)
\[-516.5\text{ }cal/mol\] done
clear
View Solution play_arrow
-
question_answer50)
A certain reaction is non spontaneous at 298K. The entropy change during the reaction is\[121\text{ }J{{K}^{-1}}\]. Is the reaction is endothermic or exothermic? The minimum value of \[\Delta H\] for the reaction is
A)
endothermic, \[\Delta H=36.06\text{ }kJ\] done
clear
B)
exothermic, \[\Delta H=-36.06\text{ }kJ\] done
clear
C)
endothermic, \[\Delta H=60.12\text{ }kJ\] done
clear
D)
exothermic, \[\Delta H=-60.12\text{ }kJ\] done
clear
View Solution play_arrow
-
question_answer51)
The entropy of a sample of a certain substance increases by \[0.836\text{ }J\text{ }{{K}^{-1}}\] on adding reversibly 0.3344 J of heat at constant temperature. The temperature of the sample is:
A)
\[2.5\text{ }K\] done
clear
B)
\[0.3\text{ }K\] done
clear
C)
\[0.016\text{ }K\] done
clear
D)
\[~0.4\text{ }K\] done
clear
View Solution play_arrow
-
question_answer52)
One mole of an ideal gas is expanded isothermally and reversibly to half of its initial pressure. \[\Delta S\] for the process in \[J\text{ }{{K}^{-1}}\text{ }mo{{l}^{-1}}\] is \[[ln2=0.693\,and\,R=8.314,J/(mol\,K)]\]
A)
6.76 done
clear
B)
5.76 done
clear
C)
10.76 done
clear
D)
8.03 done
clear
View Solution play_arrow
-
question_answer53)
The enthalpy of hydrogenation of cyclohexene is\[-119.5\text{ }kJ\text{ }mo{{l}^{-1}}\]. If resonance energy of benzene is\[-150.4\text{ }kJ\text{ }mo{{l}^{-1}}\], its enthalpy of hydrogenation would be
A)
\[-208.1kg\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-269.9\text{ }kg\text{ }mo{{l}^{-1}}\] done
clear
C)
\[-358.5\text{ }kg\text{ }mo{{l}^{-1}}\] done
clear
D)
\[-508.9\text{ }kg\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer54)
The enthalpies of formation of \[A{{l}_{2}}{{O}_{3}}\] and \[C{{r}_{2}}{{O}_{3}}\]are\[-1596\text{ }kJ\] and \[-1134\text{ }kJ\] respectively. \[\Delta H\]for the reaction \[2Al+C{{r}_{2}}{{O}_{3}}\to 2Cr+A{{l}_{2}}{{O}_{3}}\] is
A)
\[-2730\text{ }kJ\] done
clear
B)
\[-462\,kJ\] done
clear
C)
\[-1365\,kJ\] done
clear
D)
\[+2730\,kJ\] done
clear
View Solution play_arrow
-
question_answer55)
The favourable conditions for a spontaneous reaction are
A)
\[T\Delta S>\Delta H,\Delta H=+ve,\text{ }\Delta S=+ve\] done
clear
B)
\[T\Delta S>\Delta H,\text{ }\Delta H=+ve,\text{ }\Delta S=-ve\] done
clear
C)
\[T\Delta S=\Delta H,\text{ }\Delta H=-ve,\text{ }\Delta S=-ve\] done
clear
D)
\[T\Delta S=\Delta H,\text{ }\Delta H=+ve,\,\,\Delta S=+ve\] done
clear
View Solution play_arrow
-
question_answer56)
The standard Gibb's free energy change, \[\Delta G{}^\circ \] is related to equilibrium constant, \[{{K}_{P}}\] as
A)
\[{{K}_{P}}=-RT\] In \[\Delta {{G}^{{}^\circ }}\] done
clear
B)
\[{{K}_{P}}={{\left[ \frac{e}{RT} \right]}^{\Delta {{G}^{{}^\circ }}}}\] done
clear
C)
\[{{K}_{P}}=-\frac{\Delta G}{RT}\] done
clear
D)
\[{{K}_{P}}={{e}^{-\Delta {{G}^{{}^\circ }}/RT}}\] done
clear
View Solution play_arrow
-
question_answer57)
Which of the following salts should cause maximum cooling when 1 mole of it is dissolved in the same volume of water?
A)
\[NaCl;\text{ }\Delta {{H}^{{}^\circ }}=5.35\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[KN{{O}_{3}};\text{ }\Delta {{H}^{{}^\circ }}=53.5\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[KOH;\Delta {{H}^{{}^\circ }}=-56.0\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
D)
\[HBr;\Delta {{H}^{{}^\circ }}=-83.3\text{ }kJmo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer58)
The molar enthalpies of combustion of isobutene and n-butane are \[-2870\text{ }kJ\text{ }mo{{l}^{-1}}\] and \[-2875\text{ }kJ\text{ }mo{{l}^{-1}}\] respectively at 298 K and 1 atm. Calculate \[\Delta H{}^\circ \] for the conversion of 1 mole of n-butane to 1 mole of isobutane
A)
\[-8\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[+8\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[-5748\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
D)
\[+5748\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer59)
The enthalpy of atomisation of \[C{{H}_{4}}\] and \[{{C}_{2}}{{H}_{6}}\] are 360 and \[620\text{ }kcal\text{ }mo{{l}^{-1}}\] respectively. The \[C-C\] bond energy is expected to be
A)
\[210\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
B)
\[~80\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
C)
\[130\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
D)
\[180\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer60)
Boron can undergo the following reactions with the given enthalpy changes: \[2B\left( s \right)+\frac{3}{2}{{O}_{2}}\left( g \right)\xrightarrow{{}}{{B}_{2}}{{O}_{3}}\left( s \right);\text{ }\Delta H=-1260\text{ }kJ\]\[2B(s)+3{{H}_{2}}(g)\xrightarrow{{}}{{B}_{2}}{{H}_{6}}(g);\Delta H=30kJ\] Assume no other reactions are occurring. If in a container (operating at constant pressure) which is isolated from the surrounding, mixture of \[{{H}_{2}}\](gas) and \[{{O}_{2}}\] (gas) are passed over excess of B(s), then calculate the molar ratio \[({{O}_{2}}:H)\] so that temperature of the container do not change :
A)
15 : 3 done
clear
B)
42 : 1 done
clear
C)
1 : 42 done
clear
D)
1 : 84 done
clear
View Solution play_arrow
-
question_answer61)
Combustion of sucrose is used by aerobic organisms for providing energy for the life sustaining processes. If all the capturing of energy from the reaction is done through electrical process (non P-V work) then calculate maximum available energy which can be captured by combustion of 34.2 g of sucrose Given: \[\Delta {{H}_{combustion}}\left( sucrose \right)=-\text{ }6000\text{ }kJ\text{ }mo{{l}^{-1}}\] \[\Delta {{S}_{combustion}}=180J/Kmol\] and body temperature is 300 K
A)
600 kJ done
clear
B)
594.6 kJ done
clear
C)
5.4 kJ done
clear
D)
605.4 kJ done
clear
View Solution play_arrow
-
question_answer62)
The factor of \[\Delta G\] values is important in metallurgy. The \[\Delta G\] values for the following reactions at \[{{800}^{{}^\circ }}C\] are given as:
| \[{{S}_{2}}\left( s \right)+2{{O}_{2}}\left( g \right)\xrightarrow{{}}2S{{O}_{2}}\left( g \right);\Delta G=-544kJ\] |
| \[2Zn\left( s \right)+{{S}_{2}}\left( s \right)\xrightarrow{{}}2ZnS\left( s \right);\Delta G=-293\text{ }kJ\] |
| \[2Zn\left( s \right)+{{O}_{2}}\left( g \right)\xrightarrow{{}}2ZnO\left( s \right)\text{ };\Delta G=-480kJ\] |
| Then \[\Delta G\] for the reaction: |
| \[2ZnS(s)+3{{O}_{2}}(g)\xrightarrow{{}}2ZnO(s)+2S{{O}_{2}}(g)\]will be: |
A)
\[-357\text{ }kJ\] done
clear
B)
\[-731kJ\] done
clear
C)
\[-773\text{ }kJ\] done
clear
D)
\[-229\text{ }kJ\] done
clear
View Solution play_arrow
-
question_answer63)
1 gram equivalent of \[{{H}_{2}}S{{O}_{4}}\] is treated with 112 g of \[KOH\] for complete neutralization. Which of the following statements is correct?
A)
13.7 kcal of heat is evolved with the formation of 87 g of \[{{K}_{2}}S{{O}_{4}}\], leaving no KOH. done
clear
B)
27.4 kcal of heat is evolved with the formation of 87 g of \[{{K}_{2}}S{{O}_{4}}\], leaving 4 gram equivalent of KOH. done
clear
C)
15.7 kcal of heat is evolved with the formation of 1 gram equivalent of\[{{K}_{2}}S{{O}_{4}}\], leaving 56 g of KOH. done
clear
D)
13.7 kcal of heat is evolved with the formation of 87g of \[{{K}_{2}}S{{O}_{4}}\], leaving 1 gram equivalent of KOH. done
clear
View Solution play_arrow
-
question_answer64)
From the following data AH of the following reactions
| \[C(s)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}CO(g);\] |
| \[\Delta H=-110kJ\] and |
| \[C(s)+{{H}_{2}}O(g)\xrightarrow{{}}CO(g)+{{H}_{2}}(g);\] |
| \[\Delta H=132kJ\] |
| Calculate the mole composition of the mixture of steam and oxygen on being passes over coke at 1273 K, keeping the temperature constant. |
A)
1 : 0.6 done
clear
B)
0.6 : 1 done
clear
C)
2 : 3 done
clear
D)
3 : 2 done
clear
View Solution play_arrow
-
question_answer65)
Gasoline has an enthalpy of combustion 24000 kJ/ gallon. When gasoline burns in an automobile engine, approximately 30% of the energy released is used to produce mechanical work. The remainder is lost as heat transfer to the engine's cooling system. As a start on estimating how much heat transfer is required, calculate what mass of water could be heated from \[25{}^\circ C\]to \[75{}^\circ C\] by the combustion of 1.0 gallon of gasoline in an automobile? (Given:\[C({{H}_{2}}O)=4.18J/{{g}^{{}^\circ }}C\])
A)
34.45 kg done
clear
B)
80.383 kg done
clear
C)
22 kg done
clear
D)
224 kg done
clear
View Solution play_arrow
-
question_answer66)
2 moles of an ideal gas at \[27{}^\circ C\]temperature is expanded reversibly from 2 L to 20 L. Find entropy change (R = 2 cal/mol K)
A)
92.1 done
clear
B)
0 done
clear
C)
4 done
clear
D)
9.2 done
clear
View Solution play_arrow
-
question_answer67)
One mole of solid iron was vaporized in an oven at its boiling point of 3433 K and enthalpy of vaporization of iron is \[344.3\text{ }kJ\text{ }mo{{l}^{-1}}\]. The value of entropy vaporization (in J \[mo{{l}^{-1}}\]) of iron is
A)
100 done
clear
B)
10 done
clear
C)
\[-100\] done
clear
D)
110 done
clear
View Solution play_arrow
-
question_answer68)
A reaction is spontaneous at low temperature but non-spontaneous at high temperature. Which of the following is true for the reaction?
A)
\[\Delta H>0,\,\Delta S>0\] done
clear
B)
\[\Delta H<0,\text{ }\Delta S>0\] done
clear
C)
\[\Delta H>0,\,\Delta S=0\] done
clear
D)
\[\Delta H<0,\,\Delta S<0\] done
clear
View Solution play_arrow
-
question_answer69)
Which of the following pairs of processes is certain to occur in a spontaneous chemical reaction?
A)
Exothermic and increasing disorder done
clear
B)
Exothermic and decreasing disorder done
clear
C)
Endothermic and increasing disorder done
clear
D)
Endothermic and decreasing disorder done
clear
View Solution play_arrow
-
question_answer70)
For the reaction \[2{{C}_{6}}{{H}_{5}}C{{O}_{2}}H\left( s \right)+15{{O}_{2}}\left( g \right)\xrightarrow{{}}\]\[14C{{O}_{2}}\left( g \right)+6{{H}_{2}}O\left( g \right)\] \[\Delta U{}^\circ =-772.7\text{ }kJ\text{ }mo{{l}^{-1}}\] at 298 K. Calculate \[\Delta H{}^\circ \]
A)
\[+760.3\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-760.3\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[+670.3\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
D)
\[-790.3\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer71)
\[\Delta H_{f}^{0}\] of water is\[-285.8\text{ }kJ\text{ }mo{{l}^{-1}}\]. If enthalpy of neutralisation of monoacidic strong base is \[-57.3\text{ }kJmo{{l}^{-1}}\]. \[\Delta H_{f}^{0}\] of \[O{{H}^{-}}\] ion will be
A)
\[-114.25\text{ }kJ\,mo{{l}^{-1}}\] done
clear
B)
\[114.25\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[228.5\text{ }kJ\,mo{{l}^{-1}}\] done
clear
D)
\[-228.5\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer72)
The heat of atomization of \[P{{H}_{3}}(g)\] is \[228\text{ }kcal\text{ }mo{{l}^{-1}}\] and that of \[{{P}_{2}}{{H}_{4}}(g)\] is\[335\text{ }kcal\text{ }mo{{l}^{-1}}\]. The energy of the P-P bond is
A)
\[102\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
B)
\[51\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
C)
\[26\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
D)
\[204\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer73)
For the reaction \[CO(g)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g),\Delta H\], and \[\Delta S\] are \[-283\text{ }kJ\] and \[-87\text{ }J{{K}^{-1}}\], respectively. It was intended to carry out this reaction at 1000, 1500, 3000 and 3500 K. At which of these temperatures would this reaction be thermodynamically spontaneous?
A)
1500 and 3500 K done
clear
B)
3000 and 3500 K done
clear
C)
1000, 1500 and 3000 K done
clear
D)
1500, 3000 and 3500 K done
clear
View Solution play_arrow
-
question_answer74)
The molar entropies of HI (g) and I (g) at 298 K are 206.5, 114.6, and \[180.7\text{ }J\text{ }mo{{l}^{-1}}{{K}^{-1}}\] respectively. Using the\[\Delta G{}^\circ \]given Below, calculate the bond energy of HI. \[HI\left( g \right)\xrightarrow{{}}H\left( g \right)+I\left( g \right);\text{ }\Delta G{}^\circ =271.8\text{ }kJ\]
A)
\[282.4\text{ }kJmo{{l}^{-1}}\] done
clear
B)
\[298.3\text{ }kJmo{{l}^{-1}}\] done
clear
C)
\[290.1\text{ }kJmo{{l}^{-1}}\] done
clear
D)
\[315.4\text{ }kJmo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer75)
What is the equilibrium constant if ATP hydrolysis by water produce standard free energy of \[-50\text{ }kJ/mol\] under normal body conditions?
A)
\[2.66\times {{10}^{8}}\] done
clear
B)
\[5.81\times {{10}^{8}}\] done
clear
C)
\[1.18\times {{10}^{7}}\] done
clear
D)
\[1.98\times {{10}^{8}}\] done
clear
View Solution play_arrow
-
question_answer76)
0.5 mole each of two ideal gases \[A\left( {{C}_{v,m}}=\frac{5}{2}R \right)\]and \[B({{C}_{v,m}}=3R)\] are taken in a container and expanded reversibly and adiabatically, during this process temperature of gaseous mixture decreased from 350 K to 250 K. Find \[\Delta H\] (in cal/mol) for the process:
A)
\[-100R\] done
clear
B)
\[-137.5\text{ }R\] done
clear
C)
\[-375R\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer77)
What is the free energy change \['\Delta G'\] when 1.0 mole of water at \[100{}^\circ C\] and 1 atm pressure is converted into steam at \[100{}^\circ C\] and 1 atm. pressure
A)
540 cal done
clear
B)
\[-9800\text{ }cal\] done
clear
C)
9800 cal done
clear
D)
0 cal done
clear
View Solution play_arrow
-
question_answer78)
In an irreversible process taking place at constant T and P and in which only pressure-volume work is being done, the change in Gibbs free energy (dG) and change in entropy (dS), satisfy the criteria
A)
\[{{(\Delta S)}_{V,E}}>0,{{(\Delta G)}_{T,P}}<0\] done
clear
B)
\[{{(\Delta S)}_{V,E}}=0,{{(\Delta G)}_{T,P}}=0\] done
clear
C)
\[{{(\Delta S)}_{V,E}}=0,{{(\Delta G)}_{T,P}}>0\] done
clear
D)
\[{{(\Delta S)}_{V,E}}<0,{{(\Delta G)}_{T,P}}<0\] done
clear
View Solution play_arrow
-
question_answer79)
For the auto-ionization of water at \[25{}^\circ C,\] \[{{H}_{2}}O(l)\rightleftharpoons {{H}^{+}}(aq)+O{{H}^{-}}(aq)\] equilibrium constant is \[{{10}^{-14}}\]. What is \[\Delta G{}^\circ \] for the process?
A)
\[\simeq 8\times {{10}^{4}}J\text{ }mo{{l}^{-1}}\] done
clear
B)
\[\simeq 3.5\times {{10}^{4}}Jmo{{l}^{-1}}\] done
clear
C)
\[\simeq 2\times {{10}^{4}}Jmo{{l}^{-1}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer80)
For which of the following process, \[\Delta S\] is negative?
A)
\[{{H}_{2}}\left( g \right)\xrightarrow{{}}2H\left( g \right)\] done
clear
B)
\[2S{{O}_{3}}\left( g \right)\xrightarrow{{}}2S{{O}_{2}}\left( g \right)+{{O}_{2}}\left( g \right)\] done
clear
C)
\[{{N}_{2}}(4l)\xrightarrow{compressed}{{N}_{2}}(2l)\] done
clear
D)
\[C\left( diamond \right)\xrightarrow{{}}C\left( graphite \right)\] done
clear
View Solution play_arrow
-
question_answer81)
What is the entropy change (in\[J{{K}^{-1}}\,mo{{l}^{-1}}\]) when one mole of ice is converted into water at \[0{}^\circ C\]? (The enthalpy change for the conversion of ice to liquid water is \[6.0\text{ }kJ\text{ }mo{{l}^{-1}}\]at\[0{}^\circ C\])
A)
21.98 done
clear
B)
20.13 done
clear
C)
2.013 done
clear
D)
2.198 done
clear
View Solution play_arrow
-
question_answer82)
Temperature of 5 moles of a gas is decreased by 2K at constant pressure. Indicate the correct statement
A)
Work done by gas is = 5 R done
clear
B)
Work done over the gas is = 10 R done
clear
C)
Work done by the gas = 10 R done
clear
D)
Work done =0 done
clear
View Solution play_arrow
-
question_answer83)
An ideal gas occuping a volume of \[2d{{m}^{3}}\] and a pressure of 5 bar undergoes isothermal and irreversible expansion against external pressure of 1 bar. The final volume of the system and the work involved in the process is
A)
\[10\text{ }d{{m}^{3}},1000\text{ }J\] done
clear
B)
\[8\text{ }d{{m}^{3}},\text{ }-800\text{ }J\] done
clear
C)
\[10d{{m}^{3}},-800\text{ }J\] done
clear
D)
\[10d{{m}^{3}},-1000J\] done
clear
View Solution play_arrow
-
question_answer84)
A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant temperature of\[37.0{}^\circ C\]. As it does so, it absorbs 208 J of heat. The values of q and w for the process will be: \[\left( R=8.314\text{ }J/mol\text{ }K \right)\left( \text{ln }7.5=2.01 \right)\]
A)
\[q=+208\,J,w=-208J\] done
clear
B)
\[q=-208\,J,w=-208J\] done
clear
C)
\[q=-208\,J,w=+208J\] done
clear
D)
\[q=+208J,w=+208J\] done
clear
View Solution play_arrow
-
question_answer85)
The heats of neutralisation of \[C{{H}_{3}}COOH\], \[HCOOH,\text{ }HCN\] and \[{{H}_{2}}S\] are\[-13.2,-13.4\], \[~-2.9\] and \[-3.8\text{ }kcal\] per equivalent respectively. Arrange the acids in increasing order of strength
A)
\[HCOOH>C{{H}_{3}}COOH>{{H}_{2}}S>HCN\] done
clear
B)
\[C{{H}_{3}}COOH>HCOOH>{{H}_{2}}S>HCN\] done
clear
C)
\[{{H}_{2}}S>HCOOH>C{{H}_{3}}COOH>HCN\] done
clear
D)
\[HCOOH>{{H}_{2}}S>C{{H}_{3}}COOH>HCN\] done
clear
View Solution play_arrow
-
question_answer86)
The enthalpy of neutralisation of a weak acid in 1 M solution with a strong base is \[-56.1\text{ }kcal\text{ }mo{{l}^{-1}}.\] If the enthalpy of ionisation of acid is \[1.5\text{ }kcal\text{ }mo{{l}^{-1}}\] and enthalpy of neutralisation of the strong acid with a strong base is \[-57.3\text{ }kJ\text{ }e{{q}^{-1}}\]. What is the % ionisation of the weak acid in molar solution (assume the acid is monobasic)
A)
25 done
clear
B)
20 done
clear
C)
15 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer87)
The free energy change for the following reactions are given below, \[{{C}_{2}}{{H}_{2}}(g)+\frac{5}{2}{{O}_{2}}(g)\to \] \[2C{{O}_{2}}\left( g \right)+{{H}_{2}}O\left( l \right);\Delta G{}^\circ =-1234kJ\] \[C\left( s \right)+{{O}_{2}}\left( g \right)\to C{{O}_{2}}\left( g \right);\text{ }\Delta G{}^\circ =-394kJ\] \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\to {{H}_{2}}O(l);\Delta {{G}^{{}^\circ }}=-237kJ\] What is the standard free energy change for the reaction \[{{H}_{2}}(g)+2C(s)\xrightarrow{{}}{{C}_{2}}{{H}_{2}}(g)\]
A)
\[-209\,kJ\] done
clear
B)
\[-2259\,kJ\] done
clear
C)
\[+2259\,kJ\] done
clear
D)
\[209\,kJ\] done
clear
View Solution play_arrow
-
question_answer88)
For the reaction \[{{C}_{3}}{{H}_{8}}(g)+5{{O}_{2}}(g)\to 3C{{O}_{2}}(g)+4{{H}_{2}}O(l)\] at constant temperature, \[\Delta H-\Delta E\] is
A)
\[-RT\] done
clear
B)
\[+RT\] done
clear
C)
\[-3\text{ }RT\] done
clear
D)
\[+3\text{ }RT\] done
clear
View Solution play_arrow
-
question_answer89)
Among the following, the intensive properties are
| (i) molar conductivity |
| (ii) electromotive force |
| (iii) resistance |
| (iv) heat capacity |
A)
(i) and (ii) done
clear
B)
(i), (ii) and (iii) done
clear
C)
(i) and (iv) done
clear
D)
(i) only done
clear
View Solution play_arrow
-
question_answer90)
What is the normal boiling point of mercury? Given: \[\Delta H_{f}^{{}^\circ }\left( Hg,l \right)=0;\text{ }S{}^\circ \left( Hg,l \right)=77.4\text{ }J/K\]-mol \[\Delta H_{f}^{{}^\circ }\left( Hg,g \right)=60.8\text{ }kJ/mol;\text{ }S{}^\circ \left( Hg,g \right)=174.4J/K-mol\]
A)
624.8 K done
clear
B)
626.8 K done
clear
C)
636.8 K done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer91)
When 1 mole of oxalic acid is treated with excess of \[NaOH\] in dilute aqueous solution, \[106kJ\] of heat is liberated. Predict the enthalpy of ionisation of the acid
A)
\[4.3\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-4.3\,kJ\,mo{{l}^{-1}}\] done
clear
C)
\[8.6\text{ }kJ\,mo{{l}^{-1}}\] done
clear
D)
\[-8.6\,kJ\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer92)
The lattice energy of solid \[NaCl\] is \[180\text{ }kcal\text{ }mo{{l}^{-1}}\] and enthalpy of solution is \[1\text{ }kcal\text{ }mo{{l}^{-1}}\] If the hydration energies of \[N{{a}^{+}}\] and \[C{{l}^{-}}\] ions are in the ratio 3 : 2, what is the enthalpy of hydration of sodium ion?
A)
\[-107.4\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
B)
\[107.4\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
C)
\[71.6\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
D)
\[-71.6\text{ }kcal\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer93)
Standard enthalpy of combustion of \[C{{H}_{3}}\] is \[-\,890\text{ }kJ\text{ }mo{{l}^{-1}}\] and standard enthalpy of vaporisation of water is\[40.5\text{ }kJ\text{ }mo{{l}^{-1}}\]. The enthalpy change of the reaction \[C{{H}_{4}}\left( g \right)+2{{O}_{2}}\left( g \right)\xrightarrow{{}}C{{O}_{2}}\left( g \right)+{{H}_{2}}O\left( g \right)\]
A)
\[-809.5\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-890\text{ }kJmo{{l}^{-1}}\] done
clear
C)
\[809\text{ }kJ\,mo{{l}^{-1}}\] done
clear
D)
\[-971\text{ }kJ\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer94)
Standard enthalpy and standard entropy changes for the oxidation of ammonia at 298 K are \[-\,\,382.64\text{ }kJ\text{ }mo{{l}^{-1}}\] and\[-45.6\text{ }J{{K}^{-1}}mo{{l}^{-1}}\], respectively Standard Gibb's energy change for the same reaction at 298 K is
A)
\[-22.1\,kJ\,mo{{l}^{-1}}\] done
clear
B)
\[-339.3\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
C)
\[-439.3\text{ }kJ\,mo{{l}^{-1}}\] done
clear
D)
\[-523.2\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer95)
\[\Delta G\] in \[A{{g}_{2}}O\to 2Ag+1/2{{O}_{2}}\] at a certain temperature is \[-10\text{ }kJ\text{ }mo{{l}^{-1}}\]. Pick the correct statement
A)
\[A{{g}_{2}}O\]decomposes to Ag and \[{{O}_{2}}\] done
clear
B)
Ag and \[{{O}_{2}}\] combines to form \[A{{g}_{2}}O\] done
clear
C)
Reaction is in equilibrium done
clear
D)
Reaction does not take place done
clear
View Solution play_arrow
-
question_answer96)
For complete combustion of ethanol, \[{{C}_{2}}{{H}_{5}}OH\left( l \right)+3{{O}_{2}}\left( g \right)\xrightarrow{{}}2C{{O}_{2}}\left( g \right)+3{{H}_{2}}O\left( l \right),\] the amount of heat produced as measured in bomb calorimeter, is \[1364.47\text{ }kJ\text{ }mo{{l}^{-1}}\] at\[25{}^\circ C\]. Assuming ideality the enthalpy of combustion, \[{{\Delta }_{c}}H\], for the reaction will be: \[\left( R=8.314\text{ }kJ\,mo{{l}^{-1}} \right)\]
A)
\[-1366.95\text{ }kJ\text{ }mo{{l}^{-1}}\] done
clear
B)
\[-1361.95\text{ }kJ\,mo{{l}^{-1}}\] done
clear
C)
\[-1460.95\text{ }kJ\,mo{{l}^{-1}}\] done
clear
D)
\[-1350.50\text{ }kJ\,mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer97)
Diborane is a potential rocket fuel which undergoes combustion according to the equation \[{{B}_{2}}{{H}_{6}}\left( g \right)+3{{O}_{2}}\left( s \right)\xrightarrow{{}}{{B}_{2}}{{O}_{3}}\left( s \right)+3{{H}_{2}}O\left( g \right)\]Calculate the enthalpy change for the combustion of diborane. Given
| (i) \[2B(s)+\frac{3}{2}{{O}_{2}}(g)\xrightarrow{{}}{{B}_{2}}{{O}_{3}}(s);\]\[\Delta H=-1273\text{ }kJ\]per mol |
| (ii) \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}{{H}_{2}}O(l);\]\[\Delta H=-286\text{ }kJ\] per mol |
| (iii) \[{{H}_{2}}O\left( l \right)\xrightarrow{{}}{{H}_{2}}O\left( g \right);\]\[\Delta H=44\text{ }kJ\]per mol |
| (iv) \[2B(s)+3{{H}_{2}}(g)\xrightarrow{{}}{{B}_{2}}{{H}_{6}}(g);\]\[\Delta H=36\text{ }kJ\]per mol |
A)
\[+2035\text{ }kJ\]per mol done
clear
B)
\[-2035\text{ }kJ\] per mol done
clear
C)
\[+2167kJ\] per mol done
clear
D)
\[-2167kJ\] per mol done
clear
View Solution play_arrow
-
question_answer98)
What is the amount of heat (in Joules) absorbed by 18 g of water initially at room temperature heated to \[100{}^\circ C\]? If 10 g of Cu is added to this water, than decrease in temperature (in Kelvin) of water was found to be? C (p,m) for water\[75.32\text{ }J/mol\text{ }K\]; C (p,m) for \[Cu=24.47\text{ }J/mol\text{ }K.\]
A)
5649,369 done
clear
B)
5544,324 done
clear
C)
5278,342 done
clear
D)
3425,425 done
clear
View Solution play_arrow
-
question_answer99)
How many molecules of ATP, undergo hydrolysis to raise the temperature of 180 kg of water which was originally at room temperature by\[1{}^\circ C\] ? C{P,m} water = 75.32 J/mol/K, \[\Delta H\left\{ P \right\}\] for ATP hydrolysis = 7 kcal/mol
A)
\[1.5\times {{10}^{25}}\] done
clear
B)
\[2.00\times {{10}^{23}}\] done
clear
C)
\[3.4\times {{10}^{25}}\] done
clear
D)
\[4.0\times {{10}^{24}}\] done
clear
View Solution play_arrow
-
question_answer100)
Consider the \[\Delta {{G}^{{}^\circ }}_{f}\] and \[\Delta {{H}^{{}^\circ }}_{f}(kJ/mol)\] for the following oxides. Which oxide can be most easily decomposed to form the metal and oxygen gas?
A)
\[ZnO\left( \Delta G{}^\circ =-318.4,\text{ }\Delta H{}^\circ =-348.3 \right)\] done
clear
B)
\[C{{u}_{2}}O\left( \Delta G{}^\circ =-146.0,\text{ }\Delta H{}^\circ =-168.8 \right)\] done
clear
C)
\[HgO\left( \Delta G{}^\circ =-58.5,\text{ }\Delta H{}^\circ =-90.8 \right)\] done
clear
D)
\[PbO\left( \Delta G{}^\circ =-187.9,\text{ }\Delta H{}^\circ =-217.3 \right)\] done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
