| The order of reactivity of the following alcohols towards cone. HCl is |
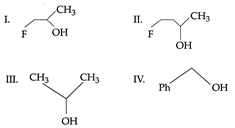 |
A) \[IV>III>II>I\]
B) \[I>II>III>IV\]
C) \[IV>III>I>II\]
D) \[I>III>II>IV\]
Correct Answer: A
Solution :
| The order of reactivity depends upon the stability of the carbocations formed. |
| Stability of carbocations is in the order. |
| \[\underset{I\,\,({{2}^{o}},\,\,F\,\,nearer)}{\mathop{FC{{H}_{2}}\overset{+}{\mathop{C}}\,HC{{H}_{3}}}}\,<\underset{II\,\,({{2}^{o}},\,\,F\,\,away)}{\mathop{FC{{H}_{2}}C{{H}_{2}}\overset{+}{\mathop{C}}\,HC{{H}_{3}}}}\,<\underset{III\,\,({{2}^{o}})}{\mathop{C{{H}_{3}}\overset{+}{\mathop{C}}\,HC{{H}_{3}}}}\,<\underset{IV\,\,(Benzyl)}{\mathop{Ph\overset{+}{\mathop{C}}\,{{H}_{2}}}}\,\] |
You need to login to perform this action.
You will be redirected in
3 sec
