| In the following compound given below the correct order of decreasing acidic strength is |
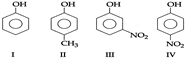 |
A) \[IV>III>I>II\]
B) \[II>I>III>IV\]
C) \[I>IV>III>II\]
D) \[III>IV>I>III\]
Correct Answer: A
Solution :
[a]| Electron-withdrawing effect of a group increases the acidity of phenols and electron-releasing group has the opposite effect. |
| In (II), there is a \[(+\,\,l)\] effect of \[C{{H}_{3}}\] group [decreases acidity] |
| In (III), there is a \[(-\,\,l)\] effect of \[N{{O}_{2}}\] group. [increases acidity] |
| In (IV), there is a \[(-\,\,l)\] and \[(-R)\] effect of \[N{{O}_{2}}\] group. [strongly increases acidity] |
You need to login to perform this action.
You will be redirected in
3 sec
