| Arrange the following compounds in order of their reactivity towards \[{{S}_{N}}2\] reaction. |
| (i) \[C{{H}_{3}}{{\left( C{{H}_{2}} \right)}_{3}}C{{H}_{2}}Br\] |
| (ii) \[{{\left( C{{H}_{3}} \right)}_{2}}CHC{{H}_{2}}C{{H}_{2}}Br\] |
| (iii) |
(iv) 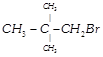 |
A) (i) > (ii) > (iii) > (iv)
B) (ii) > (iii) > (iv) > (i)
C) (iii) > (i) > (ii) > (iv)
D) (iv) > (ii) > (i) > (iii)
Correct Answer: A
Solution :
Order of reactivity of alkyl halides towards \[{{S}_{N}}2\]reaction is \[1{}^\circ \text{ }>\text{ }2{}^\circ \text{ }>\text{ }3{}^\circ \text{ }>\] aryl halides. For \[{{S}_{N}}2\] mechanism, the steric hindrance should be minimum.You need to login to perform this action.
You will be redirected in
3 sec
