| When acetone and chloroform are mixed together, which of the following observations is correct? |
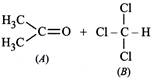 |
A) A - A and B - B interactions are stronger than A - B interactions.
B) A - A and B - B interactions are weaker than A - B interactions.
C) A - A, B - B and A - B interactions are equal.
D) The liquids form separate layers and are immiscible.
Correct Answer: B
Solution :
When acetone and chloroform are mixed together, a hydrogen bond is formed between them which increases intermolecular interactions. Hence, A - B interactions are stronger than A - A and A - B interactions.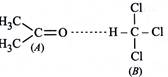
You need to login to perform this action.
You will be redirected in
3 sec
