| What is the correct observation when \[B{{r}_{2}}\] is treated with NaF, NaCl and NaI taken in three test-tubes labelled as (X), (Y) and (Z)? |
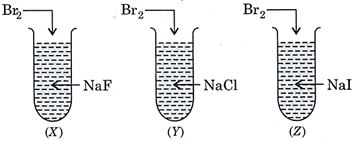 |
A) \[{{F}_{2}}\] is liberated in (X) and \[C{{l}_{2}}\] in (Y).
B) Only \[{{I}_{2}}\] is liberated in (Z).
C) Only \[C{{l}_{2}}\] is liberated in (Y).
D) Only \[{{F}_{2}}\] is liberated in (X).
Correct Answer: B
Solution :
More reactive halogen can displace less reactive halogen from its salt solution. \[{{F}_{2}}\] can displace \[C{{l}_{2}}\], \[B{{r}_{2}}\], \[{{I}_{2}}\] from their salt solutions while \[C{{l}_{2}}\] can displace \[B{{r}_{2}}\] and \[{{I}_{2}}\] from NaBr and \[NaI\,\,.\,\,B{{r}_{2}}\] can displace only \[{{I}_{2}}\] from NaI. \[2NaI+B{{r}_{2}}\to 2NaBr+{{I}_{2}}\]You need to login to perform this action.
You will be redirected in
3 sec
