| The unit cell shown in the figure belongs to |
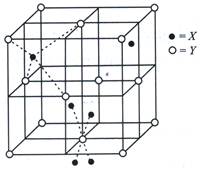 |
A) NaCl type
B) ZnS type
C) CsCl type
D) \[Ca{{F}_{2}}\]type.
Correct Answer: B
Solution :
The unit cell is ZnS type where \[{{S}^{2-}}\]ions are present at the corners and at the center of each face. \[Z{{n}^{2+}}\] ions occupy half of the tetrahedral sites.You need to login to perform this action.
You will be redirected in
3 sec
