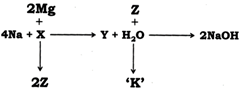
 |
| X | Y | Z | K |
| (a) \[{{H}_{2}}O\] | \[{{O}_{2}}\] | \[MgO\] | \[Mg{{(OH)}_{2}}\] |
| (b) \[{{O}_{2}}\] | \[N{{a}_{2}}{{O}_{2}}\] | \[Mg\] | \[MgO\] |
| (c) \[{{O}_{2}}\] | \[N{{a}_{2}}O\] | \[MgO\] | \[Mg{{(OH)}_{2}}\] |
| (d) \[{{H}_{2}}O\] | \[{{O}_{2}}\] | \[Mg{{(OH)}_{2}}\] | \[MgO\] |
Answer:
We know that Metallic hydroxides are formed when metal oxides are dissolved in water. \[\therefore \]\[N{{a}_{2}}O(Y)+{{H}_{2}}O\to 2NaOH\Rightarrow Y=N{{a}_{2}}O\] \[N{{a}_{2}}O\]is formed when sodium reacts with oxygen. \[\therefore \]\[4Na+{{O}_{2}}(X)\to 2N{{a}_{2}}O\Rightarrow X={{O}_{2}}\] \[\therefore \]\[2Mg+{{O}_{2}}\to 2MgO(Z)\Rightarrow Z=MgO\] \[\therefore \] \[MgO+{{H}_{2}}O\to Mg(OH)(K)\Rightarrow K=Mg{{(OH)}_{2}}\]
You need to login to perform this action.
You will be redirected in
3 sec
