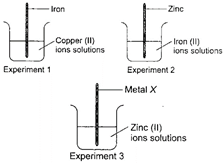
| Aditya, a class 8 student investigated the reactivity of four metals, iron, copper, zinc and an unknown metal X. He arranged three experimental set-ups as shown in the diagram and observed the changes carefully. |
|
| If reactions occur in all the three beakers then, the metal X is |
A) Cu
B) Mg
C) Fe
D) Au
Correct Answer: B
Solution :
Metal X should be more reactive than Zn to displace Zn from \[\text{ZnS}{{\text{O}}_{\text{4}}}\] solution. Among the metals given, Mg is more reactive than Zn while Cu, Fe and Au are less reactive than Zn.You need to login to perform this action.
You will be redirected in
3 sec
