| Arrange the following in the increasing order of reactivity with \[N{{H}_{3}}\]: |
| (I) \[C{{H}_{2}}O\] |
| (II) \[C{{H}_{3}}CHO\] |
| (III) \[C{{H}_{3}}-CO-C{{H}_{3}}\] |
(IV) 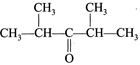 |
A) I<II<III<IV
B) III<IV<I<II
C) IV<III<II<I
D) II<I<IV<III
Correct Answer: C
Solution :
[c] Reactivity follows the ease with which a nucleophile can attack at carbonyl carbon. Greater the steric hindrance at carbonyl carbon, smaller the reactivity.You need to login to perform this action.
You will be redirected in
3 sec
