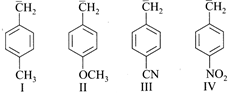
A) IV>III>I>II
B) III>IV>II>I
C) I>III>II>IV
D) II>I>III>IV
Correct Answer: A
Solution :
[a] \[-N{{O}_{2}}\] Has greater electron withdrawing power than \[-CN\]by resonance effect, hence IV is most stable followed by III. II is least stable as delocalization of negative charge is opposed by electron donating resonance effect of methoxy group.You need to login to perform this action.
You will be redirected in
3 sec
