A) The order of their reducing strength is \[{{H}_{3}}P{{O}_{2}}>{{H}_{3}}P{{O}_{3}}>{{H}_{3}}P{{O}_{4}}\]
B) the hybridisation of phosphorus is \[s{{p}^{3}}\] in all these.
C) all have one P = O
D) all of these
Correct Answer: D
Solution :
[d]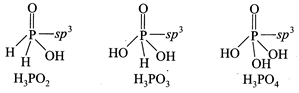
You need to login to perform this action.
You will be redirected in
3 sec
