A) \[NC{{l}_{3}}\], HCl and \[{{N}_{2}}\], \[N{{H}_{4}}Cl\]
B) \[{{N}_{2}}\], \[N{{H}_{4}}Cl\] and \[NC{{l}_{3}}\], HCl
C) \[{{N}_{2}}\], HCl and \[{{N}_{2}}\], \[N{{H}_{4}}Cl\]
D) \[{{N}_{2}}\], \[N{{H}_{4}}Cl\] and \[NC{{l}_{3}}\], HCl
Correct Answer: B
Solution :
[b] When ammonia is in excess: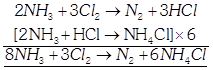 When chlorine is in excess: \[N{{H}_{3}}+HCl\to NC{{l}_{3}}+3HCl\]
When chlorine is in excess: \[N{{H}_{3}}+HCl\to NC{{l}_{3}}+3HCl\]
You need to login to perform this action.
You will be redirected in
3 sec
