question_answer 1)
If P, V, T represent pressure, volume and temperature of the gas, the correct representation of Boyle's law is [BIT Ranchi 1988]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 2)
At constant temperature, in a given mass of an ideal gas [CBSE PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 3)
Air at sea level is dense. This is a practical application of [Kerala CEE 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 4)
If 20\[c{{m}^{3}}\] gas at 1 atm. is expanded to 50 \[c{{m}^{3}}\] at constant T, then what is the final pressure [CPMT 1988]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
Which of the following statement is false [BHU 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
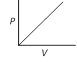
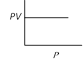
question_answer 6)
Which of the following graphs represent Boyle's law
A)
B)
C)
D)
View Solution play_arrow
question_answer 7)
Densities of two gases are in the ratio 1 : 2 and their temperatures are in the ratio 2 : 1, then the ratio of their respective pressures is [BHU 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
At constant pressure, the volume of fixed mass of an ideal gas is directly proportional to [EAMCET 1985]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
Which of the following expression at constant pressure represents Charle's law [AFMC 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
Use of hot air balloons in sports and meteorological obsevations is an application of [Kerala MEE 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
A 10 g of a gas at atmospheric pressure is cooled from \[{{273}^{o}}C\] to \[{{0}^{o}}C\] keeping the volume constant, its pressure would become
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
Pressure remaining the same, the volume of a given mass of an ideal gas increases for every degree centigrade rise in temperature by definite fraction of its volume at [CBSE PMT 1989]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
A certain sample of gas has a volume of 0.2 litre measured at 1 atm. pressure and \[{{0}^{o}}C\]. At the same pressure but at \[{{273}^{o}}C\], its volume will be [EAMCET 1992, 93; BHU 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 14)
400 \[c{{m}^{3}}\] of oxygen at\[{{27}^{o}}C\]were cooled to \[-{{3}^{o}}C\] without change in pressure. The contraction in volume will be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
The pressure p of a gas is plotted against its absolute temperature T for two different constant volumes, \[{{V}_{1}}\] and \[{{V}_{2}}\]. When \[{{V}_{1}}>{{V}_{2}}\], the
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
Two closed vessels of equal volume containing air at pressure \[{{P}_{1}}\] and temperature \[{{T}_{1}}\] are connected to each other through a narrow tube. If the temperature in one of the vessels is now maintained at \[{{T}_{1}}\] and that in the other at \[{{T}_{2}}\], what will be the pressure in the vessels
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 17)
?One gram molecule of a gas at N.T.P. occupies 22.4 litres.? This fact was derived from [CPMT 1981, 1995]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
In a closed flask of 5 litres, 1.0 g of \[{{H}_{2}}\] is heated from 300 to 600 K. which statement is not correct [CBSE PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 19)
Which one of the following statements is false [Manipal PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 20)
The compressibility of a gas is less than unity at STP. Therefore [IIT 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 21)
In the equation of sate of an ideal gas \[PV=nRT\], the value of the universal gas constant would depend only on [KCET 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 22)
In the ideal gas equation, the gas constant R has the dimensions of [NCERT 1982]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 23)
In the equation \[PV=nRT\], which one cannot be the numerical value of R [BIT 1987]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 24)
Which one of the following indicates the value of the gas constant R [EAMCET 1989]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 25)
The constant R is [Orissa 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 26)
Select one correct statement. In the gas equation, \[PV=nRT\] [CBSE PMT 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 27)
The correct value of the gas constant R is close to [CBSE PMT 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 28)
S.I. unit of gas constant R is [CPMT 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 29)
Gas equation \[PV=nRT\] is obeyed by [BHU 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 30)
For an ideal gas number of moles per litre in terms of its pressure P, gas constant R and temperature T is [AIEEE 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 31)
If two moles of an ideal gas at 546 K occupy a volume of 44.8 litres, the pressure must be [NCERT 1981; JIPMER 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 32)
How many moles of He gas occupy 22.4 litres at \[{{30}^{o}}C\] and one atmospheric pressure [KCET 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 33)
Volume of 0.5 mole of a gas at 1 atm. pressure and \[273K\] is [EAMCET 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 34)
At \[{{0}^{o}}C\] and one atm pressure, a gas occupies 100 cc. If the pressure is increased to one and a half-time and temperature is increased by one-third of absolute temperature, then final volume of the gas will be [DCE 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 35)
Correct gas equation is [CBSE PMT 1989; CPMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 36)
Two separate bulbs contain ideal gases A and B. The density of gas A is twice that of gas B. The molecular mass of A is half that of gas B. The two gases are at the same temperature. The ratio of the pressure of A to that of gas B is [BHU 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 37)
16 g of oxygen and 3 g of hydrogen are mixed and kept at 760 mm pressure and \[{{0}^{o}}C\]. The total volume occupied by the mixture will be nearly [Vellore CMC 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 38)
Pure hydrogen sulphide is stored in a tank of 100 litre capacity at \[{{20}^{o}}C\] and 2 atm pressure. The mass of the gas will be [CPMT 1989]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 39)
At N.T.P. the volume of a gas is found to be 273 ml. What will be the volume of this gas at 600 mm Hg and \[{{273}^{o}}C\] [CPMT 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 40)
One litre of a gas weighs 2 g at 300 K and 1 atm pressure. If the pressure is made 0.75 atm, at which of the following temperatures will one litre of the same gas weigh one gram [CBSE PMT 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 41)
A wheather balloon filled with hydrogen at 1 atm and \[{{27}^{o}}C\] has volume equal to 12000 litres. On ascending it reaches a place where the temperature is \[-{{23}^{o}}C\] and pressure is 0.5 atm. The volume of the balloon is [CBSE PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 42)
The density of a gas at \[{{27}^{o}}C\] and 1 atm is d. Pressure remaining constant at which of the following temperatures will its density become 0.75 d [CBSE PMT 1992]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 43)
A sample of gas occupies 100 ml at \[{{27}^{o}}C\] and 740 mm pressure. When its volume is changed to 80 ml at 740 mm pressure, the temperature of the gas will be [Vellore CMC 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 44)
The total pressure exerted by a number of non-reacting gases is equal to the sum of the partial pressures of the gases under the same conditions is known as [CPMT 1986]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 45)
?Equal volumes of all gases at the same temperature and pressure contain equal number of particles.? This statement is a direct consequence of [Kerala MEE 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 46)
If three unreactive gases having partial pressures \[{{P}_{A}},\,{{P}_{B}}\] and \[{{P}_{C}}\] and their moles are 1, 2 and 3 respectively then their total pressure will be [CPMT 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 47)
Dalton's law of partial pressure will not apply to which of the following mixture of gases [Bihar MADT 1981]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 48)
Which of the following mixtures of gases does not obey Dalton's law of partial pressure [CBSE PMT 1996: Kerala PMT 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 49)
To which of the following gaseous mixtures is Dalton's law not applicable
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 50)
Equal amounts of two gases of molecular weight 4 and 40 are mixed. The pressure of the mixture is 1.1 atm. The partial pressure of the light gas in this mixture is [CBSE PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 51)
Rate of diffusion of a gas is [IIT 1985; CPMT 1987]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 52)
Which of the following gas will have highest rate of diffusion [Pb. CET Sample paper 1993; CPMT 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 53)
Which of the following relationship is correct, where r is the rate of diffusion of a gas and d is its density [CPMT 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 54)
According to Grahman's law at a given temperature, the ratio of the rates of diffusion \[{{r}_{A}}/{{r}_{B}}\] of gases A and B is given by [IIT 1998]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 55)
The ratio of the rate of diffusion of a given element to that of helium is 1.4. The molecular weight of the element is [Kerala PMT 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 56)
A gas diffuse 1/5 times as fast as hydrogen. Its molecular weight is [CPMT 1992; Bihar CEE 1982]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 57)
The molecular weight of a gas which diffuses through a porous plug at 1/6th of the speed of hydrogen under identical conditions is [EAMCET 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 58)
Molecular weight of a gas that diffuses twice as rapidly as the gas with molecular weight 64 is [EAMCET 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 59)
The densities of hydrogen and oxygen are 0.09 and 1.44 g \[{{L}^{-1}}\]. If the rate of diffusion of hydrogen is 1 then that of oxygen in the same units will be [RPMT 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 60)
If rate of diffusion of A is 5 times that of B, what will be the density ratio of A and B [AFMC 1994]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 61)
The densities of two gases are in the ratio of 1 : 16. The ratio of their rates of diffusion is [CPMT 1995]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 62)
At constant volume and temperature conditions, the rate of diffusion \[{{D}_{A}}\] and \[{{D}_{B}}\] of gases A and B having densities \[{{\rho }_{A}}\] and \[{{\rho }_{B}}\] are related by the expression [IIT 1993]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 63)
Atmolysis is a process of
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 64)
A bottle of ammonia and a bottle of dry hydrogen chloride connected through a long tube are opened simultaneously at both ends, the white ammonium chloride ring first formed will be [IIT 1988]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 65)
Which of the following pairs will diffuse at the same rate through a porous plug [EAMCET 1990]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 66)
If 4 g of oxygen diffuse through a very narrow hole, how much hydrogen would have diffused under identical conditions [CPMT 1971]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 67)
A gas diffuse at a rate which is twice that of another gas B. The ratio of molecular weights of A to B is [EAMCET 1986]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 68)
Two grams of hydrogen diffuse from a container in 10 minutes. How many grams of oxygen would diffuse through the same container in the same time under similar conditions [MNR 1980]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 69)
The rate of diffusion of methane at a given temperature is twice that of X. The molecular weight of X is [MNR 1995; Kerala CEE 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 70)
X ml of \[{{H}_{2}}\] gas effuses through a hole in a container in 5 seconds. The time taken for the effusion of the same volume of the gas specified below under identical condition is [IIT 1996]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 71)
At what temperature, the rate of effusion of \[{{N}_{2}}\] would be 1.625 times that of \[S{{O}_{2}}\] at \[{{50}^{o}}C\] [CBSE PMT 1996]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 72)
Given the reaction \[C(s)+{{H}_{2}}O(l)\to CO(g)+{{H}_{2}}(g)\] calculate the volume of the gases produced at STP from 48.0 g of carbon
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 73)
4.4 g of a gas at STP occupies a volume of 2.24 L, the gas can be [Haryana CEET 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 74)
Under what conditions will a pure sample of an ideal gas not only exhibit a pressure of 1 atm but also a concentration of 1 mole \[litre-1\] \[(R=0.082\,litre\,atm\,mo{{l}^{-1}}\,{{\deg }^{-1}})\] [CBSE PMT 1993]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 75)
There are \[6.02\times {{10}^{22}}\] molecules each of \[{{N}_{2}},\,{{O}_{2}}\] and \[{{H}_{2}}\] which are mixed together at 760 mm and 273 K. The mass of the mixture in grams is [Pb. PMT 1997]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 76)
Volume of 4.4 g of \[C{{O}_{2}}\] at NTP is [Pb. CET 1997]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 77)
The energy of an ideal gas depends only on its
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 78)
A bottle of cold drink contains 200 ml liquid in which \[C{{O}_{2}}\] is 0.1 molar. Suppose \[C{{O}_{2}}\] behaves like an ideal gas, the volume of the dissolved \[C{{O}_{2}}\] at STP is [CBSE PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 79)
The vapour density of a gas is 11.2. The volume occupied by 11.2 g of this gas at N.T.P. is [MNR 1982; CBSE PMT 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 80)
A pre-weighed vessel was filled with oxygen at N.T.P. and weighted. It was then evacuated, filled with \[S{{O}_{2}}\] at the same temperature and pressure, and again weighted. The weight of oxygen will be [NCERT 1989]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 81)
Five grams each of the following gases at \[{{87}^{o}}C\] and 750 mm pressure are taken. Which of them will have the least volume [MNR 1991]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 82)
Who among the following scientists has not done any important work on gases [Bihar MADT 1980]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 83)
A closed vessel contains equal number of nitrogen and oxygen molecules at a pressure of P mm. If nitrogen is removed from the system then the pressure will be [MP PMT 1985]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 84)
If the four tubes of a car are filled to the same pressure with \[{{N}_{2}},\,{{O}_{2}},\,{{H}_{2}}\] and Ne separately, then which one will be filled first [Manipal PMT 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 85)
Which of the following gas mixture is not applicable for Dalton?s law of partial pressure [Pb. CET 2002]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 86)
At what pressure a quantity of gas will occupy a volume of \[60\,ml\], if it occupies a volume of \[100ml\] at a pressure of \[720mm\]? (while temperature is constant) : [Pb. CET 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 87)
At constant temperature and pressure which gas will diffuse first \[{{H}_{2}}\] or \[{{O}_{2}}\]? [Pb. CET 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 88)
When a jar containing gaseous mixture of equal volumes of \[C{{O}_{2}}\] and \[{{H}_{2}}\] is placed in a solution of sodium hydroxide, the solution level will [Pb. CET 2001]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 89)
At S.T.P. \[1g\,CaC{{O}_{3}}\] on decomposition gives \[C{{O}_{2}}\] [Pb. CET 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 90)
At NTP, the density of a gas, whose molecular weight is 45 is [Pb. CET 2001, 03]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 91)
What is the ratio of diffusion rate of oxygen and hydrogen [Pb. CET 2003]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 92)
The maximum number of molecules is present in [CBSE PMT 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 93)
One litre oxygen gas at STP will weigh [Pb. CET 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 94)
How will you separate mixture of two gases [AFMC 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 95)
The rate of diffusion of hydrogen gas is [MH CET 2003; Pb. CET 2000]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 96)
Hydrogen diffuses six times faster than gas \[A\]. The molar mass of gas \[A\] is [KCET 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 97)
At what pressure will a quantity of gas, which occupies \[100\,ml\] at a pressue of \[720\,mm\], occupy a volume of \[84\,ml\] [DPMT 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 98)
Containers \[A\] and \[B\] have same gases. Pressure, volume and temperature of \[A\] are all twice that of \[B,\] then the ratio of number of molecules of \[A\] and \[B\] are [AFMC 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 99)
A mixture of \[N{{O}_{2}}\] and \[{{N}_{2}}{{O}_{4}}\] has a vapour density of 38.3 at \[300K\]. What is the number of moles of \[N{{O}_{2}}\] in \[100g\] of the mixture [Kerala PMT 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
E)
done
clear
View Solution play_arrow
question_answer 100)
A cylinder of 5 litres capacity, filled with air at NTP is connected with another evacuated cylinder of 30 litres of capacity. The resultant air pressure in both the cylinders will be [BHU 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 101)
A certain mass of gas occupies a volume of 300 c.c. at 27C and 620 mm pressure. The volume of this gas at \[{{47}^{o}}C\] and 640 mm pressure will be [MH CET 2003]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 102)
What will be the volume of the mixture after the reaction? \[\underset{\text{4 litre}}{\mathop{N{{H}_{3}}}}\,+\underset{\text{1}\text{.5 litre}}{\mathop{HCl}}\,\to \underset{\text{(solid)}}{\mathop{N{{H}_{4}}Cl}}\,\] [BVP 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 103)
The pressure and temperature of \[4d{{m}^{3}}\] of carbon dioxide gas are doubled. Then the volume of carbon dioxide gas would be [KCET 2004]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 104)
If the absolute temperature of an ideal gas become double and pressure become half, the volume of gas would be [Kerala CET 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
E)
done
clear
View Solution play_arrow
question_answer 105)
At what temperature, the sample of neon gas would be heated to double of its pressure, if the initial volume of gas is/are reduced to 15% at \[{{75}^{o}}C\] [Kerala CET 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
E)
done
clear
View Solution play_arrow
question_answer 106)
Equation of Boyle's law is [DPMT 2005]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
