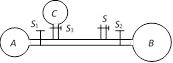
A) B will start collapsing with volumes of A and C increasing
B) C will start collapsing with volumes of A and B increasing
C) C and A both will start collapsing with the volume of B increasing
D) Volumes of A, B and C will become equal at equilibrium
Correct Answer: C
Solution :
Excess pressure inside soap bubble is inversely proportional to the radius of bubble i.e. \[\Delta P\propto \frac{1}{r}\] This means that bubbles A and C posses greater pressure inside it than B. So the air will move from A and C towards B.You need to login to perform this action.
You will be redirected in
3 sec
