A) Less reactive than benzyl chloride
B) More reactive than ethyl bromide
C) Nearly as reactive as methyl chloride
D) More reactive than isopropyl chloride
Correct Answer: A
Solution :
Chlorobenzene In chlorobenzene the lone pairs present on Cl atom get involved in resonance with \[\pi \]electrons of benzene due to which \[C-Cl\] bond acquires double bond character Hence, reactivity decreases.
In chlorobenzene the lone pairs present on Cl atom get involved in resonance with \[\pi \]electrons of benzene due to which \[C-Cl\] bond acquires double bond character Hence, reactivity decreases. 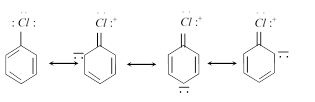 \[\underset{\text{more reactive}}{\mathop{{{C}_{2}}{{H}_{5}}-Cl}}\,>\underset{\text{less reactive}}{\mathop{{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-Cl}}\,\]
\[\underset{\text{more reactive}}{\mathop{{{C}_{2}}{{H}_{5}}-Cl}}\,>\underset{\text{less reactive}}{\mathop{{{C}_{6}}{{H}_{5}}-C{{H}_{2}}-Cl}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
