A) Pentene-1
B) cis pentene-2
C) trans pentene-2
D) 2-ethoxypentane
Correct Answer: C
Solution :
\[C{{H}_{3}}-\underset{Br\,\,}{\mathop{\underset{|\,\,\,}{\mathop{CH}}\,}}\,-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}+KOH\] \[\underset{\begin{smallmatrix} \text{Elimination} \\ \,\,\,\,\,\text{reaction} \end{smallmatrix}}{\mathop{\xrightarrow{{{C}_{2}}{{H}_{5}}OH}}}\,\underset{\text{2-Pentene}\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{C{{H}_{3}}-CH=CH-C{{H}_{2}}-C{{H}_{3}}}}\,\] When alkyl halide reacts with alc. KOH then it favours elimination reaction (Dehydrohalogenation). Since, Trans pentene-2 is more symmetrical than cis isomers. Hence, it is main product.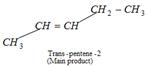
You need to login to perform this action.
You will be redirected in
3 sec
