A) \[{{C}_{3}}{{H}_{7}}N{{H}_{2}}\]
B) \[N{{H}_{3}}\]
C) \[C{{H}_{3}}N{{H}_{2}}\]
D) \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\]
E) \[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\]
Correct Answer: D
Solution :
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] is least basic compound due to resonance by which the Lone pair of nitrogen takes part in resonance & due to unavailability of lone pair on N Aniline become less basic. The Lone pair of N is delocalized into benzene ring by resonance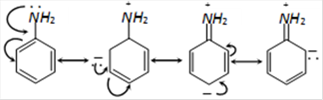
You need to login to perform this action.
You will be redirected in
3 sec
