A) Acetone
B) Acetophenone
C) Acetic acid
D) Acetyl acetone
Correct Answer: D
Solution :
The amount of enolic form is highest (76%) in acetyl acetone because keto group is a much better electron-withdrawing group. \[\overset{{}}{\longleftrightarrow}\overset{O\,....\,H\,\,O\,\,\,\,\,}{\mathop{\overset{|\,|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|\,\,\,\,\,\,\,\,\,}{\mathop{C{{H}_{3}}C-CH=C-C{{H}_{3}}}}\,}}\,\]
\[\overset{{}}{\longleftrightarrow}\overset{O\,....\,H\,\,O\,\,\,\,\,}{\mathop{\overset{|\,|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|\,\,\,\,\,\,\,\,\,}{\mathop{C{{H}_{3}}C-CH=C-C{{H}_{3}}}}\,}}\,\] 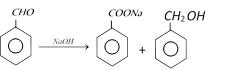
You need to login to perform this action.
You will be redirected in
3 sec
