| (I) |
(II) 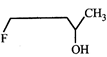 |
(III) 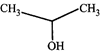 |
(IV) |
A) I>II>III>IV
B) I>III>II>IV
C) IV>III>II>I
D) IV>III>I>II
Correct Answer: C
Solution :
[c] The order of reactivity depends upon the stability of the carbocations formed. Remember that presence of electron-withdrawing roup intensifies i.e., destabilises the carbocation thus (i) and (ii) are less stable than (iii). Further (i) is less stable than (ii) because -I effect is more pronounced in (i) due to less distance between F and positive charge. Thus the stability order of the four carbocations and reactivity of their parent alcohols will be IV > III > II > I
Remember that presence of electron-withdrawing roup intensifies i.e., destabilises the carbocation thus (i) and (ii) are less stable than (iii). Further (i) is less stable than (ii) because -I effect is more pronounced in (i) due to less distance between F and positive charge. Thus the stability order of the four carbocations and reactivity of their parent alcohols will be IV > III > II > I
You need to login to perform this action.
You will be redirected in
3 sec
