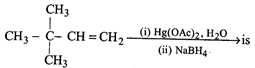
A) \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\underset{OH}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\]
B) \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\underset{{}}{\mathop{\underset{{}}{\mathop{C}}\,}}\,{{H}_{2}}-C{{H}_{2}}OH\]
C) \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\overset{OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{C}}\,}}\,H-C{{H}_{3}}\]
D) \[HOC{{H}_{2}}-\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-\underset{{}}{\mathop{\underset{{}}{\mathop{C}}\,}}\,{{H}_{2}}-C{{H}_{3}}\]
Correct Answer: A
Solution :
[a] Recall that oxymercuratoin-reduction of alkenes lead to hydration in Markovnikov's way. Thus \[{{(C{{H}_{3}})}_{3}}CCH=C{{H}_{2}}\xrightarrow[(ii)\,NaB{{H}_{4}}]{(i)\,Hg{{(OAc)}_{2}}.{{H}_{2}}O}{{(C{{H}_{3}})}_{3}}C\overset{OH}{\mathop{\overset{|}{\mathop{C}}\,}}\,HC{{H}_{3}}\]You need to login to perform this action.
You will be redirected in
3 sec
