A) \[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\]
B) \[C{{H}_{3}}C{{H}_{2}}N\vec{=}C\]
C) \[C{{H}_{3}}C\equiv N\]
D) \[C{{H}_{2}}C{{H}_{2}}OH\]
Correct Answer: B
Solution :
[b]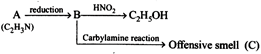 Given reactions indicate that B has \[1{}^\circ N{{H}_{2}}\] group, and thus \[A,{{C}_{2}}{{H}_{3}}N\], should be \[C{{H}_{3}}C\equiv N.\] Hence C should be \[C{{H}_{3}}C{{H}_{2}}NC\] \[\underset{A}{\mathop{C{{H}_{3}}C\equiv N}}\,\xrightarrow{reaction}\underset{B}{\mathop{C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}}}\,\xrightarrow[KOH]{CHC{{l}_{3}}}\] \[\underset{C}{\mathop{C{{H}_{3}}C{{H}_{2}}N}}\,\vec{=}C\]
Given reactions indicate that B has \[1{}^\circ N{{H}_{2}}\] group, and thus \[A,{{C}_{2}}{{H}_{3}}N\], should be \[C{{H}_{3}}C\equiv N.\] Hence C should be \[C{{H}_{3}}C{{H}_{2}}NC\] \[\underset{A}{\mathop{C{{H}_{3}}C\equiv N}}\,\xrightarrow{reaction}\underset{B}{\mathop{C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}}}\,\xrightarrow[KOH]{CHC{{l}_{3}}}\] \[\underset{C}{\mathop{C{{H}_{3}}C{{H}_{2}}N}}\,\vec{=}C\]
You need to login to perform this action.
You will be redirected in
3 sec
