A) atomic size of boron is larger than nitrogen
B) Boron is metal while nitrogen is gas
C) \[B-F\] bonds are non-polar while \[N-F\] bonds are polar
D) \[B{{F}_{3}}\] is planar but \[N{{F}_{3}}\] is pyramidal
Correct Answer: D
Solution :
[d] The shape of \[B{{F}_{3}}\] is trigonal planar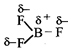 and \[\mu =0\] hence it is non polar. The shape of \[N{{F}_{3}}\] is pyramidal
and \[\mu =0\] hence it is non polar. The shape of \[N{{F}_{3}}\] is pyramidal You need to login to perform this action.
You will be redirected in
3 sec
