A) \[s{{p}^{3}}\]
B) \[s{{p}^{2}}\]
C) sp
D) \[ds{{p}^{2}}\]
Correct Answer: A
Solution :
[a]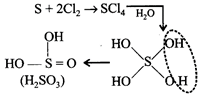 Hybridization of \[{{H}_{2}}S{{O}_{3}}=\frac{1}{2}(6+2+0)=4=s{{p}^{3}}\]
Hybridization of \[{{H}_{2}}S{{O}_{3}}=\frac{1}{2}(6+2+0)=4=s{{p}^{3}}\]
You need to login to perform this action.
You will be redirected in
3 sec
