A) geometry, magnetic moment
B) geometry, hybridization
C) magnetic moment, colour
D) hybridization, number of d-electrons
Correct Answer: C
Solution :
[c] \[F{{e}^{2+}}in{{\left[ Fe{{({{H}_{2}}O)}_{6}} \right]}^{2+}}\]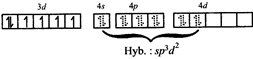 Colour: Pale green\[\mu =4.9\text{ }B.M\].; octahedral \[F{{e}^{2+}}in{{\left[ Fe{{(CN)}_{6}} \right]}^{4-}}\]
Colour: Pale green\[\mu =4.9\text{ }B.M\].; octahedral \[F{{e}^{2+}}in{{\left[ Fe{{(CN)}_{6}} \right]}^{4-}}\] 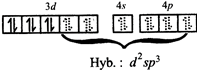 Colour: Yellow;\[\mu =0\]; octahedral
Colour: Yellow;\[\mu =0\]; octahedral
You need to login to perform this action.
You will be redirected in
3 sec
