A) it is a low spin complex
B) it is diamagnetic
C) it shows geometrical isomerism
D) [a] and [b] both
Correct Answer: D
Solution :
[d] Complex X is\[{{[Fe{{(en)}_{3}}]}^{2+}}\]; as 'en' is a strong field ligand pairing of electrons will take place.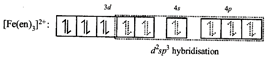 Hence, hybridisation is \[{{d}^{2}}s{{p}^{3}}\] and complex is diamagnetic. As it has 3 bidentate symmetrical 'en' ligands so it will not show geometrical isomerism.
Hence, hybridisation is \[{{d}^{2}}s{{p}^{3}}\] and complex is diamagnetic. As it has 3 bidentate symmetrical 'en' ligands so it will not show geometrical isomerism.
You need to login to perform this action.
You will be redirected in
3 sec
