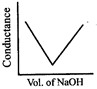
A)
B)
C)
D)
Correct Answer: A
Solution :
[a] Molar conductivity of \[{{H}^{+}}\] and \[O{{H}^{-}}\] are very high as compare to other ions. Initially conductance of solution sharply decreases due to consumption of free \[{{H}^{+}}\] After complete neutralization further slightly increases due to presence of\[O{{H}^{-}}\].You need to login to perform this action.
You will be redirected in
3 sec
