A) 2-Ethoxypentane
B) Pentene-1
C) cis-Pentene-2
D) trans-Pentene-2
Correct Answer: D
Solution :
[d] Potassium ethoxide is a strong base, hence causes elimination reaction. The alkene formed is governed by Saytzeffrule "more substituted an alkene, higher will be its stability". \[C{{H}_{3}}\overset{Br}{\mathop{\overset{|}{\mathop{C}}\,}}\,HC{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\xrightarrow{^{-}O{{C}_{2}}{{H}_{5}}}\] \[C{{H}_{2}}=\underset{Pentene-1}{\mathop{CHC{{H}_{2}}C{{H}_{2}}C{{H}_{3}}}}\,+C{{H}_{3}}CH\] \[\underset{Pentene\,-2\,(more\,stable)}{\mathop{=CHC{{H}_{2}}C{{H}_{3}}}}\,\] Further pentene-2 shows geometrical isomerism, cis- and trans- in which trans-isomer having bulkieri groups away from each other is more stable than the cis-isomer.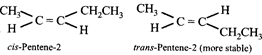
You need to login to perform this action.
You will be redirected in
3 sec
