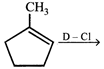
 Where D is an isotope of hydrogen
Where D is an isotope of hydrogen
A)
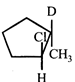
B)
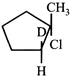
C)
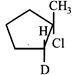
D)
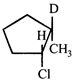
Correct Answer: B , C
Solution :
(b, c)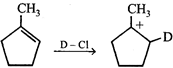 Formation of above species is more favourable. The stability order of carbocations is
Formation of above species is more favourable. The stability order of carbocations is 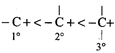 Now, carbocation formed is \[s{{p}^{2}}\] hybridised that is triangular planar as shown below
Now, carbocation formed is \[s{{p}^{2}}\] hybridised that is triangular planar as shown below 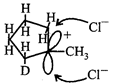 Either the
Either the 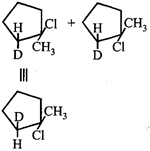
Solution :
(b, c) Formation of above species is more favourable. The stability order of carbocations is
Formation of above species is more favourable. The stability order of carbocations is 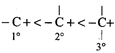 Now, carbocation formed is \[s{{p}^{2}}\] hybridised that is triangular planar as shown below
Now, carbocation formed is \[s{{p}^{2}}\] hybridised that is triangular planar as shown below  Either the
Either the 
You need to login to perform this action.
You will be redirected in
3 sec
