A)
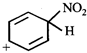
B)

C)
![]()
D) All of the three
Correct Answer: C
Solution :
[c] An aromatic compound have cyclic clouds of delocalised \[\left( 4n+2 \right)\pi \] electrons above and below the plane of the molecule. Among the given three compounds, only compound [c] satisfies these conditions.| |
|
|
| Carbon bearing \[-N{{O}_{2}}\] (or \[-C{{H}_{3}}\]) group is \[s{{p}^{3}}\] hybridised and does not have a p orbital with the result delocalisation of \[\pi \] electrons over the complete ring is interrupted. | 4\[\pi \] electrons and 2 non-bonding electrons (present in p orbtial of N) form a cyclic cloud of 6\[\pi \] electrons |
You need to login to perform this action.
You will be redirected in
3 sec
