(I)  |
(II) 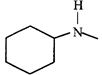 |
(III) 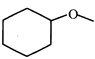 |
(IV) 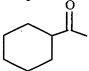 |
A) (I)
B) (II)
C) (III)
D) (IV)
Correct Answer: B
Solution :
[b] When the given mixture is shaken with 1 M\[HCl\], amine get protonated and becomes cation \[\left( RNH_{2}^{\oplus } \right),\] which does not dissolve in organic solvent but usually dissolve in \[{{H}_{2}}O\] due to its charge. So, shaking with aqueous \[HCl\] will pull amines into the aqueous phase and leave all other compounds in organic layer.You need to login to perform this action.
You will be redirected in
3 sec
