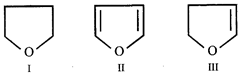
A) \[I>III>II\]
B) \[III>II>I\]
C) \[II>III>I\]
D) All are equally soluble
Correct Answer: A
Solution :
[a] Higher the electron density on O, stronger is the H-bond with water and thus more is the solubility. Thus solubility of the three ethers follow the order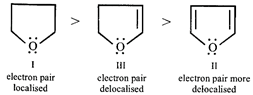
You need to login to perform this action.
You will be redirected in
3 sec
