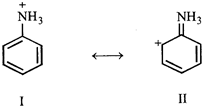
A) Structure II is not acceptable because carbonium ions are less stable than ammonium ions
B) II is not acceptable because it is non- aromatic
C) II is not acceptable because here nitrogen has 10 valence electrons
D) II is an acceptable canonical structure.
Correct Answer: C
Solution :
[c] N has no d orbital, hence it can't accommodate more than 8 electrons in its valence shell.You need to login to perform this action.
You will be redirected in
3 sec
