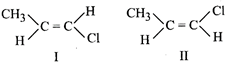
A)
Dipole moment
B.P.
M.P.
Stability
I>II
I>II
II>I
I>II
B)
Dipole moment
B.P.
M.P.
Stability
II>I
II>I
II>I
II>I
C)
Dipole moment
B.P.
M.P.
Stability
I>II
I>II
I>II
I>II
D)
Dipole moment
B.P.
M.P.
Stability
II>I
II>I
I>II
I>II
Correct Answer: C
Solution :
[c] In compounds I has more dipole moment than II, hence its boiling point will be higher. Melting point depends on symmetry, therefore I has higher melting point than II. Steric crowding in II is more than in I therefore I is more stable than II.
I has more dipole moment than II, hence its boiling point will be higher. Melting point depends on symmetry, therefore I has higher melting point than II. Steric crowding in II is more than in I therefore I is more stable than II.
You need to login to perform this action.
You will be redirected in
3 sec
