A)
![]()
B)
![]()
C)
![]()
D)
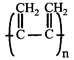
Correct Answer: D
Solution :
[d] In polymerisation of 1,3-butadiene either 1,4-polymerisation or 1,2-polymerisation occurs. In case of 1,4-polymerisation, the double bond shifts at \[{{C}_{2}}\] and \[{{C}_{4}}\] carbon, while the chain propagates from \[{{C}_{1}}\] and \[{{C}_{4}}\] end. In this either trans or cis polymeric chain is formed. Option [a] and [b] represent 'trans' and 'cis' 1,4 polymerisation respectively. Option [c] resembles 1, 2 polymerisation, where as option [d] most unlikely to happen.You need to login to perform this action.
You will be redirected in
3 sec
