A) Heating \[AlC{{l}_{3}}.6{{H}_{2}}O\]
B) By passing dry \[HCl\] over hot aluminium powder
C) By passing dry \[C{{l}_{2}}\] over hot aluminium powder
D) By passing dry \[C{{l}_{2}}\] over a hot mixture of alumina and coke
Correct Answer: A
Solution :
[a] \[2Al+6HCl\xrightarrow{\Delta .air}2AlC{{l}_{3}}+3{{H}_{2}}\] \[2Al+3C{{l}_{2}}\xrightarrow{{}}2AlC{{l}_{3}}\]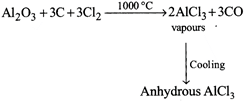 \[AlC{{l}_{3}}.6{{H}_{2}}O\xrightarrow{\Delta }Al{{\left( OH \right)}_{3}}+3HCl+3{{H}_{2}}O\] Thus \[AlC{{l}_{3}}\] cannot be obtained by this method.
\[AlC{{l}_{3}}.6{{H}_{2}}O\xrightarrow{\Delta }Al{{\left( OH \right)}_{3}}+3HCl+3{{H}_{2}}O\] Thus \[AlC{{l}_{3}}\] cannot be obtained by this method.
You need to login to perform this action.
You will be redirected in
3 sec
