A) no oxygen of a \[SiO_{4}^{4-}\] 'tetrahedron is shared with another \[SiO_{4}^{4-}\] tetrahedron
B) one oxygen of a \[SiO_{4}^{4-}\] tetrahedron is shared with another \[SiO_{4}^{4-}\] tetrahedron
C) two oxygen of a \[SiO_{4}^{4-}\] tetrahedron are shared with another \[SiO_{4}^{4-}\] tetrahedron
D) three oxygen of a \[SiO_{4}^{4-}\] tetrahedron are shared with another \[SiO_{4}^{4-}\] tetrahedron
Correct Answer: D
Solution :
[d]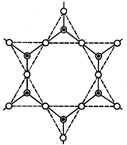 Total No. oxygen atoms per silicon atom \[=\frac{1}{2}+\frac{1}{2}+\frac{1}{2}+1=2.5\] \[\therefore \] Formula \[S{{i}_{2}}O_{5}^{2-}.\]
Total No. oxygen atoms per silicon atom \[=\frac{1}{2}+\frac{1}{2}+\frac{1}{2}+1=2.5\] \[\therefore \] Formula \[S{{i}_{2}}O_{5}^{2-}.\]
You need to login to perform this action.
You will be redirected in
3 sec
