A) \[NO_{3}^{-}\]
B) \[SO_{3}^{2-}\]
C) \[BO_{3}^{3-}\]
D) \[CO_{3}^{2-}\]
Correct Answer: B
Solution :
[b] \[p\pi -d\pi \] bonding is present in \[SO_{3}^{2-}\], N, B, C have no vacant d atomic arbitals.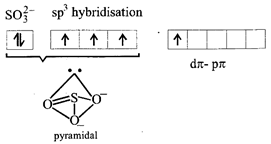
You need to login to perform this action.
You will be redirected in
3 sec
