A) \[Ca{{(HC{{O}_{3}})}_{2}}\]
B) \[CaC{{O}_{3}}\]
C) \[N{{a}_{2}}C{{O}_{3}}\]
D) \[{{K}_{2}}C{{O}_{3}}\]
Correct Answer: B
Solution :
[b] The given properties coincide with \[CaC{{O}_{3}}\]
The given properties coincide with \[CaC{{O}_{3}}\] 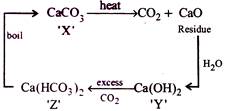
You need to login to perform this action.
You will be redirected in
3 sec
